肠道菌群是人消化道中与人体共生的微生物群落,菌群中不同种类之间以及菌群与宿主之间维持一种互惠互利的动态平衡,在正常情况下菌群结构相对稳定,对宿主不致病[1-2]。但当肠道菌群紊乱失调时,这种平衡被打破,导致多种疾病发生,如肥胖、糖尿病等代谢性疾病[3]以及阿尔茨海默症(Alzheimer’s disease,AD)等认知功能障碍疾病[4]。
膳食结构对肠道菌群的组成和结构有着重要影响,并且食物中的一些成分对某些菌属的丰度或功能有着直接影响。比如高脂饮食(high-fat diet,HFD)小鼠肠道中双歧杆菌科完全消失[5],而水产品中富含的n-3脂肪酸可增强双歧杆菌对肠壁的黏附力并显著增加双歧杆菌的数量[6]。牛磺酸是机体内的一种含硫半必需氨基酸,以游离形式广泛分布[7],具有抗氧化、调节能量代谢、抗衰老等重要生物学功能[8]。临床和流行病学研究显示,肥胖和糖尿病患者尿液和血浆中牛磺酸水平下降[9-10],而这些疾病同时都存在肠道菌群紊乱[3]。食物是体内牛磺酸的重要来源,尤其是海鲜类食物,研究发现食用富含牛磺酸海鲜较多的人患代谢综合征和心血管疾病的风险较低[11-13]。现有研究显示,牛磺酸及其与胆汁酸的结合物通过与肠道微生物相互作用,对机体的生理过程发挥着重要调节作用。
因此,本文以牛磺酸和肠道微生物为对象,阐述肠道菌群概况、体内牛磺酸的来源与转化、牛磺酸与肠道菌群的相互作用,以及牛磺酸干预对肠道菌群稳态的重塑作用。旨在深入阐释牛磺酸与肠道菌群的关系,为更有效地开发与菌群失调相关疾病的预防和治疗措施提供科学依据。
1 人体肠道菌群概况
一个70 kg体质量的人肠道内定植大约3.8×1013 个细菌,主要包括厚壁菌门(Firmicutes,60%~65%)、拟杆菌门(Bacteroidetes,20%~25%)、变形菌门(Proteobacteria,5%~10%)和放线菌门(Actinobacteria,3%)[14]。厚壁菌门主要有芽孢杆菌纲(常见菌属如芽孢杆菌属、葡萄球菌属、乳杆菌属、乳球菌属和链球菌属)和梭菌纲(如菌属有梭菌属、瘤胃球菌属、毛螺菌属)。拟杆菌门主要有拟杆菌纲包括另枝菌属、紫单胞菌属、普雷沃氏菌属、理研菌属、Alistipes等。肠道菌群的组成、丰度和多样性与宿主人种、年龄、生存环境、饮食习惯、药物使用等密切相关[15]。
根据细菌跟人类的关系,肠道菌群可分为有益菌、中性菌和有害菌3 类。其中约70%为中性菌,主要包括大肠杆菌、链球菌等,在正常情况下对机体无害,但一旦增殖失控或从肠道转移到身体的其他部位,就会导致疾病的发生[16]。有益菌约占肠道菌群的20%,如双歧杆菌、乳酸菌,拟杆菌等。有益菌可抑制有害菌定植,也可生成短链脂肪酸、必需维生素等多种营养物质,并可分解肠道的有毒有害物质如脂多糖(lipopolysaccharide,LPS)及氧化三甲胺[16-17]。而变形杆菌、沙门氏菌等有害菌受共生菌制约保持在10%,其丰度增加就会引发多种疾病[18]。
肠道菌群在自身发酵过程中可产生大量代谢产物,如牛磺酸和胆汁酸等,它们或作为代谢反应的信号分子,或直接作为底物,影响宿主的病理生理过程[19]。
2 体内牛磺酸的来源、转运与转化
2.1 体内牛磺酸的来源
人体内牛磺酸来自内源性合成和膳食供给两条途径。内源性合成的主要器官为肝脏、脑、骨骼肌等,以半胱氨酸和蛋氨酸等含硫氨基酸为前体物质经一系列催化反应合成牛磺酸。如图1所示,人和小鼠等哺乳动物体内经3 条途径合成牛磺酸,分别为半胱亚磺酸途径(途径1)、半胱胺途径(途径2)和磺基丙氨酸途径(途径3),其中途径1为最主要的生物合成途径,途径2和3为次级合成途径[8,20]。在途径1中,合成牛磺酸的限速酶有两种,分别是半胱氨酸亚磺酸脱羧酶(cysteine sulfinic acid decarboxylase,CSAD)和半胱氨酸双加氧酶(cysteine dioxygenase,CDO),它们的活力决定了合成牛磺酸的能力;啮齿类动物合成能力最强,人的合成能力较弱,而猫则无法合成[21]。另外,不同生长阶段机体合成牛磺酸的能力也不相同,与成年人相比,新生儿由于肝脏中催化甲硫氨酸生成半胱氨酸的酶活性较低,导致无法合成足够牛磺酸,因此新生儿更加依赖从食物摄取牛磺酸[22-23]。
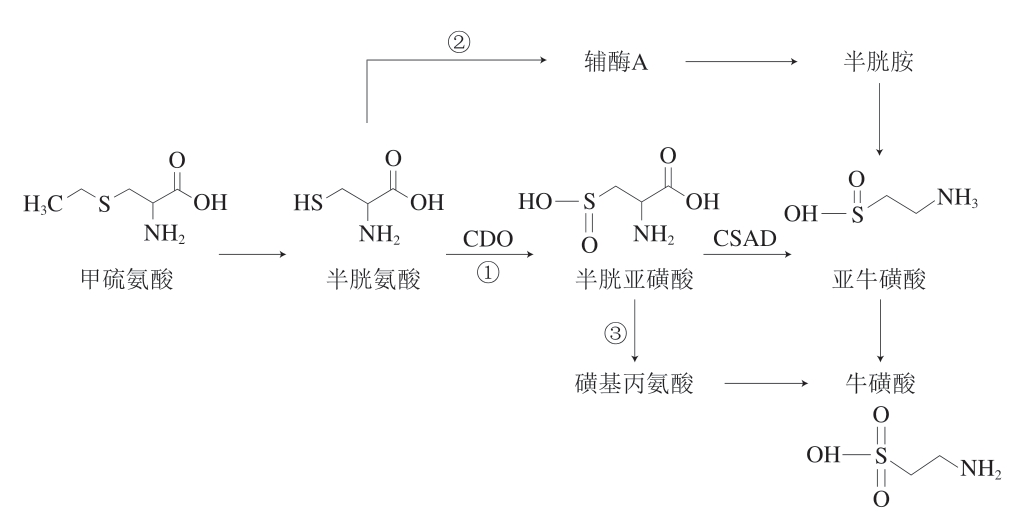
图1 牛磺酸的合成途径
Fig. 1 Synthesis pathway of taurine
一个健康成年人每天可以合成50~125 mg牛磺酸,远不能满足机体正常生理需求,因此必须经膳食补充牛磺酸[24]。目前尚没有明确的膳食牛磺酸参考摄入量[25]。除素食者等特殊人群外,成人从饮食中摄入牛磺酸的平均水平约为每天40~400 mg[25]。如表1所示,动物性食材是膳食牛磺酸的主要来源,其中水产品的牛磺酸含量最高;而植物性食物除几种藻类外其牛磺酸含量极低或几乎不含有牛磺酸[20,25-27]。在中国12 个省份的男性牛磺酸摄入情况研究中,发现北方男性的日均摄取量最低,为33.5 mg,而南方男性日均摄入量可达79.7 mg,这一差异可能源于南方地区居民较高的海鲜消费量[26]。
表1 食材牛磺酸含量[20,25-27]
Table 1 Taurine contents in foods[20,25-27]

分类食材牛磺酸含量/(mg/100 g)陆生食材鸡肉169牛肉64猪里脊肉61羊肉47猪肝42动物性食材水生产品海螺851蛤蜊520章鱼388鱿鱼356扇贝332鲫鱼205中华黄花鱼187虾143藻类红藻979坛紫菜(干)646多管藻241植物性食材其他枸杞1.13榛子0.586松子0.418杏仁0.224胡桃0.193黑豆0.115
2.2 体内牛磺酸的转运和代谢
机体内细胞膜上广泛存在牛磺酸转运蛋白,负责从膜外获取牛磺酸转运至细胞内,包括Na+/Cl-依赖型牛磺酸转运蛋白(taurine transporter,TauT)和质子偶联氨基酸转运蛋白1(proton-coupled amino acid transporter 1,PAT1)。其中,TauT对底物具有高亲和力,在牛磺酸浓度低时被激活;而PAT1对底物亲和力低但具有高容量,牛磺酸浓度较高时被激活[28-29]。TauT敲除小鼠与野生型小鼠对比,牛磺酸转运被抑制,骨骼肌和心肌中的牛磺酸水平降低了约98%,而脑、肾和肝脏中的牛磺酸水平降低了70%~90%[30-31],证明TauT是体内牛磺酸转运最主要的蛋白。敲除TauT后小鼠表现出心脏、肝脏、肾脏等组织功能改变,会导致心肌肌原纤维断裂、破坏线粒体和线粒体外膜肿胀,诱发心肌病,损害肾脏发育,并导致肝脏纤维化、非特异性肝炎和肿瘤形成[29-32],可见牛磺酸在生长发育和机体生理功能维持中的重要作用。
食物来源的牛磺酸经肠细胞膜上TauT和PAT1转运进入肠细胞后(图2),经门静脉循环进入肝脏,继而分布至全身组织,其中心脏、脑、骨骼肌等组织中浓度较高[29]。在人肝脏中,肝细胞合成的初级胆汁酸为胆酸(cholic acid,CA)和鹅脱氧胆酸(chenodeoxycholic acid,CDCA),可在胆汁酸-辅酶A连接酶(bile acid CoA-ligase,BAL)和胆汁酸酰基辅酶A:氨基酸N-酰基转移酶(bile acid CoA:amino acid N-acyltransferase,BAAT)的催化下与牛磺酸结合形成牛磺酸结合型胆汁酸(taurine-conjugated bile acids,Tau-Bas),主要包括牛磺胆酸(taurocholate acid,TCA)和牛磺鹅脱氧胆酸(taurochenodeoxycholic acid,TCDCA),成为初级胆汁酸的组成成分。结合型胆汁酸占胆汁酸池的90%[33],在成人的胆汁酸池中结合型胆汁酸主要是甘氨酸结合型,Tau-Bas仅占胆汁酸池的30%,但小鼠胆汁酸池中则有95%的胆汁酸都为Tau-Bas[34-35]。不同物种Tau-Bas分类见表2。与游离胆汁酸相比,Tau-Bas细胞毒性降低且亲水性更高[36]。肝细胞产生的胆汁酸由胆盐输出泵和多药耐药相关蛋白2转运至胆囊储存[37]。进餐后,Tau-Bas随着胆汁从胆囊释放到小肠中参与脂肪和脂溶性维生素等食物的消化吸收,并且肠道中的Tau-Bas可被肠道菌群修饰[38]。95%的胆汁酸可被重吸收进入门静脉循环,包括沿整个肠道吸收以及被回肠细胞顶端膜的钠依赖性胆汁酸转运蛋白主动吸收,然后经肝细胞表面的钠离子-牛磺胆酸共转运蛋白(Na+/taurocholate cotransporting polypeptide,NTCP)或钠非依赖性有机阴离子转运多肽(organic anion-transporting polypeptides,OATP)转运系统进入肝细胞,重新结合并分泌到胆汁中,形成肠肝循环;而其余胆汁酸经粪便排出[39-40]。牛磺酸也被证明可以通过上调肝脏胆固醇7α-羟化酶(cholesterol 7α-hydroxylase,CYP7A1)促进胆固醇向胆汁酸转化、抑制回肠对胆汁酸的重吸收,促进粪便中胆汁酸排泄等环节,从而调节胆汁酸生成和排泄[41]。
表2 不同物种Tau-Bas分类
Table 2 Classification of Tau-Bas from different species
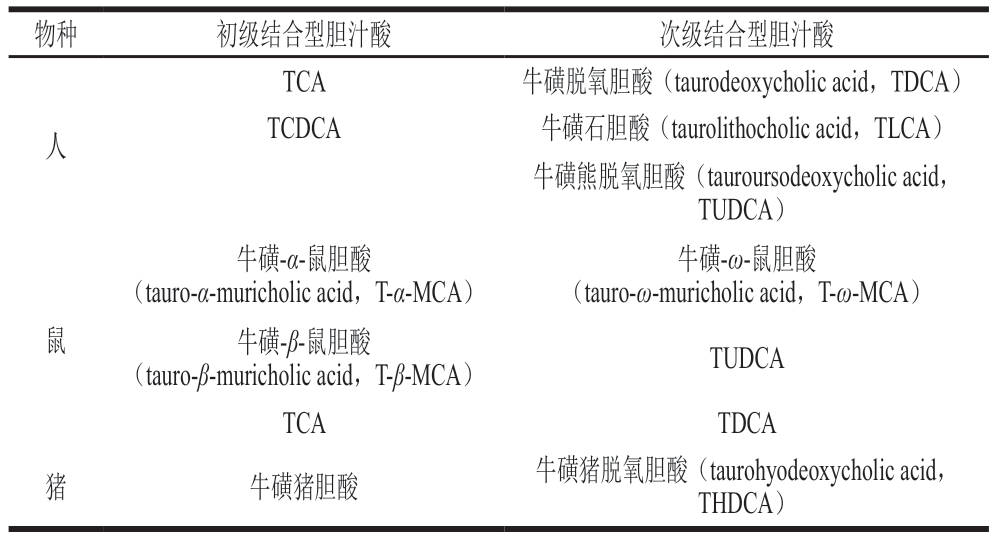
物种初级结合型胆汁酸次级结合型胆汁酸人TCA牛磺脱氧胆酸(taurodeoxycholic acid,TDCA)TCDCA牛磺石胆酸(taurolithocholic acid,TLCA)牛磺熊脱氧胆酸(tauroursodeoxycholic acid,TUDCA)牛磺-ω-鼠胆酸(tauro-ω-muricholic acid,T-ω-MCA)牛磺-β-鼠胆酸(tauro-β-muricholic acid,T-β-MCA)TUDCA TCATDCA猪牛磺-α-鼠胆酸(tauro-α-muricholic acid,T-α-MCA)鼠牛磺猪胆酸牛磺猪脱氧胆酸(taurohyodeoxycholic acid,THDCA)
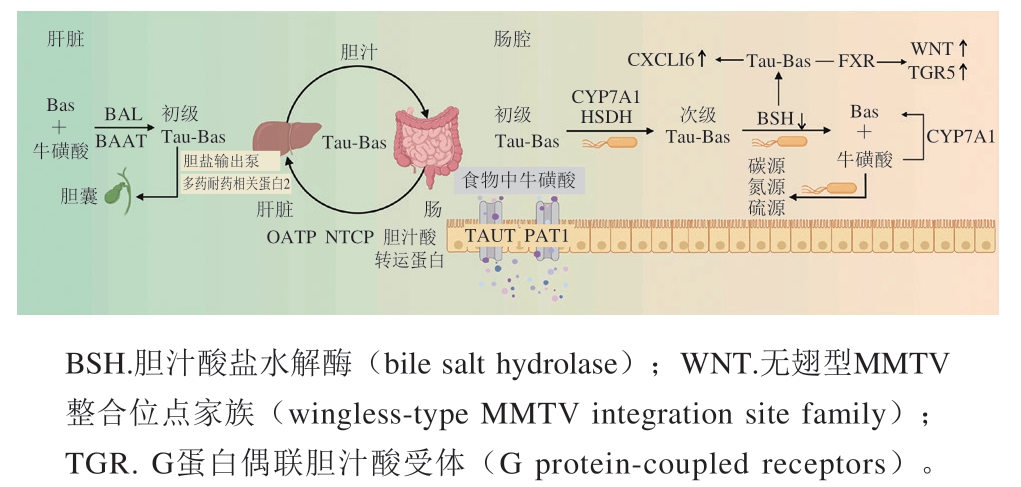
图2 牛磺酸的代谢转换途径
Fig. 2 Metabolic conversion pathway of taurine
3 牛磺酸/Tau-Bas与肠道微生物
进入肠道的Tau-Bas在肠道微生物的作用下可解离释放出牛磺酸,这是肠道牛磺酸的主要来源,而游离的牛磺酸也可被肠道微生物分解利用。肠道微生物将包括Tau-Bas的胆汁酸转化为次级胆汁酸从而增加了胆汁酸的多样性,有利于胆汁酸经粪便排出,菌群产生的次级胆汁酸量约为胆汁酸总量的5%。Tau-Bas可从肠道重吸收,充当信号分子调节宿主生理功能;反之,Tau-Bas也会影响肠道菌群的组成和数量。
3.1 肠道微生物对Tau-Bas多样性的影响
肠道微生物会对肝脏来源的初级胆汁酸进行修饰,主要包括对胆汁酸的早期解偶联、脱氢、脱羟基、差向异构化等作用。初级胆汁酸经CYP7A1脱羟基、脱氧,以及羟基类固醇脱氢酶(hydroxysteroid dehydrogenase,HSDH)的修饰下生成游离型次级胆汁酸脱氧胆酸(deoxycholic acid,DCA)、熊脱氧胆酸(ursodeoxycholic acid,UDCA)和石胆酸(litho chalic acid,LCA)以及对应的结合型次级胆汁酸[42]。目前发现拟杆菌属、梭状芽孢杆菌属、真杆菌属、乳酸菌属和埃希氏菌属的厌氧菌可以通过CYP7A1进行胆汁酸转化[43]。结直肠癌患者的粪便中出现较高浓度的7α-脱羟基梭菌,使机体肠道的次级胆汁酸含量升高,这可能是疾病发生发展的原因之一[44]。HSDH在肠道菌群中的放线菌门、变形菌门、厚壁菌门和拟杆菌门等多种微生物中表达,其异常表达会导致类固醇诱导的肥胖、糖尿病、高血压或炎症的发生[45]。
肠道中的结合型胆汁酸会在微生物释放的BSH作用下解偶联,如Tau-Bas会生成游离胆汁酸和牛磺酸,这也是牛磺酸在肠道中的主要来源[46]。人类肠道微生物群的BSH基因分类鉴定发现,591 个BSHs归属于12 个门的117 个属,包括放线菌门、拟杆菌门、厚壁菌门和变形菌门等。超过一半的BSHs属于厚壁菌门,主要包括葡萄球菌属、拟杆菌属、肠球菌属、双歧杆菌属、消化梭状芽孢杆菌属和副拟杆菌属等[47]。在超重的结直肠癌患者和HFD诱导的结直肠癌小鼠粪便中均发现表达BSH的拟杆菌丰度升高,使得结肠中未结合的胆汁酸增多,导致多余的胆汁酸逃逸到结肠中,激活WNT信号通路及TGR5,加速了结直肠癌进展[48]。而抑制富含BSH的芽孢杆菌、肠球菌等微生物和BSH活性,减少了Tau-Bas的解偶联,在高脂血症小鼠中表现出了降低胆固醇水平的效果[49]。
研究发现,无菌大/小鼠或伪无菌大/小鼠,胆汁酸多样性均减少,而肝脏中T-β-MCA和TCA以及肾脏中的TCA比例显著增加[50],无菌小鼠肝脏中游离牛磺酸浓度升高及尿液牛磺酸排泄量增高[51]。可见肠道微生物对胆汁酸特别是Tau-Bas代谢至关重要。给予小鼠抗生素后,Tau-Bas中T-ω-MCA、TDCA、TLCA和TUDCA均降低,T-β-MCA则升高,而对TCA没有明显影响[52]。Swann等[50]也发现无菌大鼠由于给予抗生素后肠道菌群丰度降低,使得初级胆汁酸T-β-MCA成为主要胆汁酸,这与菌群生成BSH减少有关。初级胆汁酸T-β-MCA可上调趋化因子CXC型趋化因子配体16(CXC chemokine ligand 16,CXCL16)基因表达,促进了自然杀伤(natural killing,NK)细胞的募集,但次级胆汁酸ω-MCA作用则与之相反[52]。使用万古霉素消灭小鼠肠道中介导初级胆汁酸到次级胆汁酸代谢的细菌革兰氏阳性菌后,明显提高了NK细胞积累,并表现出对小鼠肝脏肿瘤的抑制作用[52]。
总之,肠道微生物通过BSH和CYP7A1等活性酶对胆汁酸进行修饰代谢,增加了Tau-Bas的多样性,因此BSH和CYP7A1微生物成为了潜在的微生物靶点。作为肠道微生物重要的代谢产物,Tau-Bas可随着肠道菌群组成的改变而变化,其中初级胆汁酸的增加可促进NK细胞的募集,从而增强免疫功能,因此纠正相应的菌属或Tau-Bas是相关疾病的有效治疗策略。
3.2 Tau-Bas对法尼醇X受体(farnesoid X receptor,FXR)/TGR信号通路的影响
胆汁酸是FXR和TGR5的天然配体。FXR主要配体是初级胆汁酸,Tau-Bas中TCDCA、TCA是FXR的激活剂,T-α-MCA和T-β-MCA则是FXR拮抗剂[53-54]。TGR5主要由次级胆汁酸激活,尤其是Tau-Bas对TGR5的亲和力增强。胆汁酸通过激活FXR和TGR5,维持能量和胆汁酸稳态,并参与调控葡萄糖代谢[55-57]。
在肝脏中,CYP7A1是从胆固醇合成胆汁酸的关键酶,过量的胆汁酸激活FXR信号通路后可负反馈下调CYP7A1的表达[58],从而维持胆固醇/胆汁酸代谢平衡,也是降胆固醇的治疗靶点之一。胆汁酸还可通过激活FXR而抑制CSAD表达参与调节牛磺酸合成[59],并发现这一作用可能与肠道菌群有关[60]。研究结果显示,HFD诱导的肥胖大鼠在减重手术Roux-en-Y胃旁路术(Rouxen-Y gastric bypass,RYGB)后,肠道微生物代谢特征发生改变;进一步将RYGB的肠道微生物移植至肥胖大鼠后也出现RYGB的减肥效果,同时肠腔内BSH丰度下降,肠腔内和血中Tau-Bas均增加,并且检测到血浆及盲肠中牛磺酸水平上调[61]。这些Tau-Bas通过肠道中的FXR和靶器官中的TGR5作用于肠道及机体的多种细胞,最终诱导脂肪产热和代谢改善[61]。可见,肠道微生物在维持机体牛磺酸和Tau-Bas水平中起着关键作用,并通过FXR/TGR5信号通路调节全身代谢。
戒酒药物双硫仑可降低梭状芽孢杆菌介导的7α-脱羟基反应,抑制次级胆汁酸合成,导致TCDCA水平增加,进而激活FXR信号,最终改善非酒精性脂肪性肝炎[62]。益生菌鼠李糖乳酪杆菌GG(Lacticaseibacillus rhamnosus GG,LGG)同样改变了肠道菌群并逆转了胆管结扎(bile duct ligation,BDL)所致T-β-MCA升高,上调了肠道FXR信号通路,证明LGG可用于治疗BDL引起的小鼠肝纤维化[63]。另一方面,Li Fei等[64]发现抗氧化剂tempol可降低产生BSH的乳酸菌属细菌,从而抑制肠道FXR信号通路,最终缓解小鼠肥胖。由此可见,FXR的激动和拮抗作用在不同的情况下对机体的影响并不一致。肝脏中的FXR激活对脂肪变性有保护作用,但肠道FXR则可能促进饮食诱导的肥胖和脂肪变性。因此可能需要综合考虑FXR通路状态及肠道微生物状态,以全面评估其对健康的影响。
3.3 Tau-Bas对肠道微生物的调节
肠道微生物与上皮细胞互利共生组成了肠道屏障,共同维护肠道稳态。作为人体肠道内两大主要菌门的厚壁菌门和拟杆菌门,通常较低的厚壁菌门/拟杆菌门(Firmicutes/Bacteroidetes,F/B)比值表明肠道微环境更健康[65]。HFD饲喂的小鼠肠道拟杆菌门丰度减少而厚壁菌门丰度增加,F/B值升高,而给予TUDCA则能够显著逆转这两大门类细菌丰度的异常改变,F/B值得以恢复。并且,给予TUDCA还显著增加了抗炎细菌普拉梭菌和嗜黏蛋白阿克曼氏菌,而促炎细菌Mucispirillum和活泼瘤胃球菌则分别下降了90.9%和66.6%[65]。断奶猪仔补充TUDCA同样表现出副拟杆菌门和黏螺旋菌的相对丰度增加,有害菌链球菌和密螺旋体属丰度降低,改善了肠道屏障功能[66]。给予非胆源性胰腺炎小鼠TUDCA后,发现肠道乳酸菌属、瘤胃球菌属、普雷沃氏菌科等菌群增加,缓解了病症[67]。而石斑鱼给予TCA后肠道菌群的结构和胆汁酸组成均发生了改变,芽孢杆菌属、拟杆菌属、梭菌属、乳杆菌属等BSHs主要菌属丰度提高,胆汁酸水解增加,提高了CA和CDCA等未结合的初级胆汁酸水平[68]。这些研究证实了Tau-Bas在维护健康肠道菌群方面的重要作用,尤其是在一些肠道菌群紊乱的疾病中,给予特定的Tau-Bas可增加有益菌丰度并抑制有害菌定植的治疗潜力。
3.4 肠道微生物对解离牛磺酸的分解利用
肠道Tau-Bas解离后的游离牛磺酸可被不同种类的细菌分解,将其作为碳、硫、氮和能量来源[69]。以8 名健康人类粪便为样本,加入10 mmol/L牛磺酸进行发酵,30 h后牛磺酸仍有95%以上的残留;而给予氧气时,83%的牛磺酸会在20 h内被降解,说明牛磺酸的分解需要有氧环境[70]。研究发现好氧细菌曲霉属中的黑曲霉菌、链霉菌属、假单胞菌和大肠杆菌可代谢牛磺酸为硫源[71]。大肠杆菌染色体上的跨膜转运蛋白TauABC构成了牛磺酸的摄取系统[72],在有氧条件下参与细菌牛磺酸的摄取和脱磺化的过程[73]。TauA蛋白含有一种特定的信号肽,用于转运到细胞周质中,该蛋白几乎能捕获所有的游离牛磺酸[72]。TauB和TauC蛋白分别与ABC型转运系统的ATP结合蛋白和跨膜蛋白具有序列相似性。而TauD蛋白是一种α-酮戊二酸依赖性双加氧酶,优先从牛磺酸中释放亚硫酸盐作为硫源利用[69]。目前已在多种细菌中检测到了TauABC基因,如大肠杆菌[73]、铜绿假单胞菌[74]和枯草芽孢杆菌[75]。除了TauABC基因簇外,在红细菌属的Rhodobacter capsulatus中发现tpa-tauR-xsc基因也参与了牛磺酸的利用,它与TauABC相似,可以催化牛磺酸形成亚硫酸盐、乙酸盐等产物[76],使得红细菌可以利用牛磺酸作为唯一碳源或氮源[77-78]。除好氧细菌外,Haange等[79]研究发现厌氧菌沃氏嗜胆菌(Bilophila wadsworthii)也可分解牛磺酸作为其硫源,并与其形成负反馈回路,从而维持饥饿状态下的葡萄糖和胆固醇稳态。牛磺酸氨基转移酶是许多降解牛磺酸的细菌中催化牛磺酸分解的第1步,研究发现人类肠道中的双歧杆菌存在这种酶[80]。综上可知,牛磺酸可以被肠道细菌分解并广泛作为碳源、氮源及硫源利用。
4 牛磺酸对疾病所致肠道微生物紊乱的调节
4.1 牛磺酸/Tau-Bas水平与肠道菌群失调
多种疾病伴随着体内牛磺酸水平的下降以及Tau-Bas水平的变化,除此之外也发现这些疾病与肠道菌群失调有关。
在肥胖、动脉粥样硬化、胰腺炎、AD等疾病中,发现血、尿、粪或脑脊液的牛磺酸水平多数情况表现为下降(表3)。如AD患者脑脊液中牛磺酸水平降低25%[81],肥胖志愿者的血浆牛磺酸水平降低41%[9],抑郁症患者体内中也发现相同变化[82]。而这些疾病患者也表现出肠道菌群失调的情况。如肥胖和动脉粥样硬化患者体内都发现厚壁菌门(如金黄色葡萄球菌属、链球菌属)异常升高[83-87],而在胰腺炎、AD和自闭症谱系障碍(autistic spectrum disorder,ASD)患者中多表现为厚壁菌门下降[67,88-91]。而厚壁菌门中瘤胃球菌属表现为在未破裂颅内动脉瘤(unruptured intracranial aneurysms,UIA)患者中丰度升高[82],但在AD患者中下降[90]。拟杆菌门的细菌多数情况下是肠道有益菌,但在溢出肠道后则表现为致病特性。研究发现,ASD儿童、AD和UIA瘤患者肠道拟杆菌门都异常增加[82,89,91-92],而在肥胖、动脉粥样硬化、胰腺炎、抑郁症患者中则表现为下降[83-84,86-87,89,93-94]。Li Hao等[92]证实UIA血牛磺酸水平降低与肠道微生物有关。小鼠接受UIA患者的肠道菌群移植后,发现UIA的发生率增加,同时血清牛磺酸含量降低。进一步研究发现,给予小鼠Hungatella hathewayi可直接导致血牛磺酸下降,而补充牛磺酸则能逆转UIA的进展,这表明H. hathewayi导致的牛磺酸耗竭是引发UIA的关键因素[92]。而一项临床试验调查了66 名ASD儿童血清牛磺酸浓度,其中有21 名患儿牛磺酸水平浓度较低(<106 μmol/L),表明牛磺酸可能是ASD的有效生物标志物[95]。将ASD患者肠道微生物群移植到无菌小鼠中可促进小鼠表现出类似ASD的症状,肠道内牛磺酸含量下降了约50%,而补充牛磺酸则可缓解ASD的症状[96]。但也有相反的结果,ASD血牛磺酸升高而尿牛磺酸降低[97],具体原因尚不清楚。动脉粥样硬化心血管疾病中,微生物膜上牛磺酸转运系统的缺失可能是导致肠道微生物异常的原因之一[86]。总之,这些疾病都伴有肠道菌群失衡,其中消耗牛磺酸的菌群丰度增加可能导致牛磺酸水平下降,这可能是疾病发生发展的重要分子机制之一。
表3 肠道微生物紊乱与牛磺酸/Tau-Bas水平
Table 3 Gut microflora disorders and taurine/Tau-Bas levels
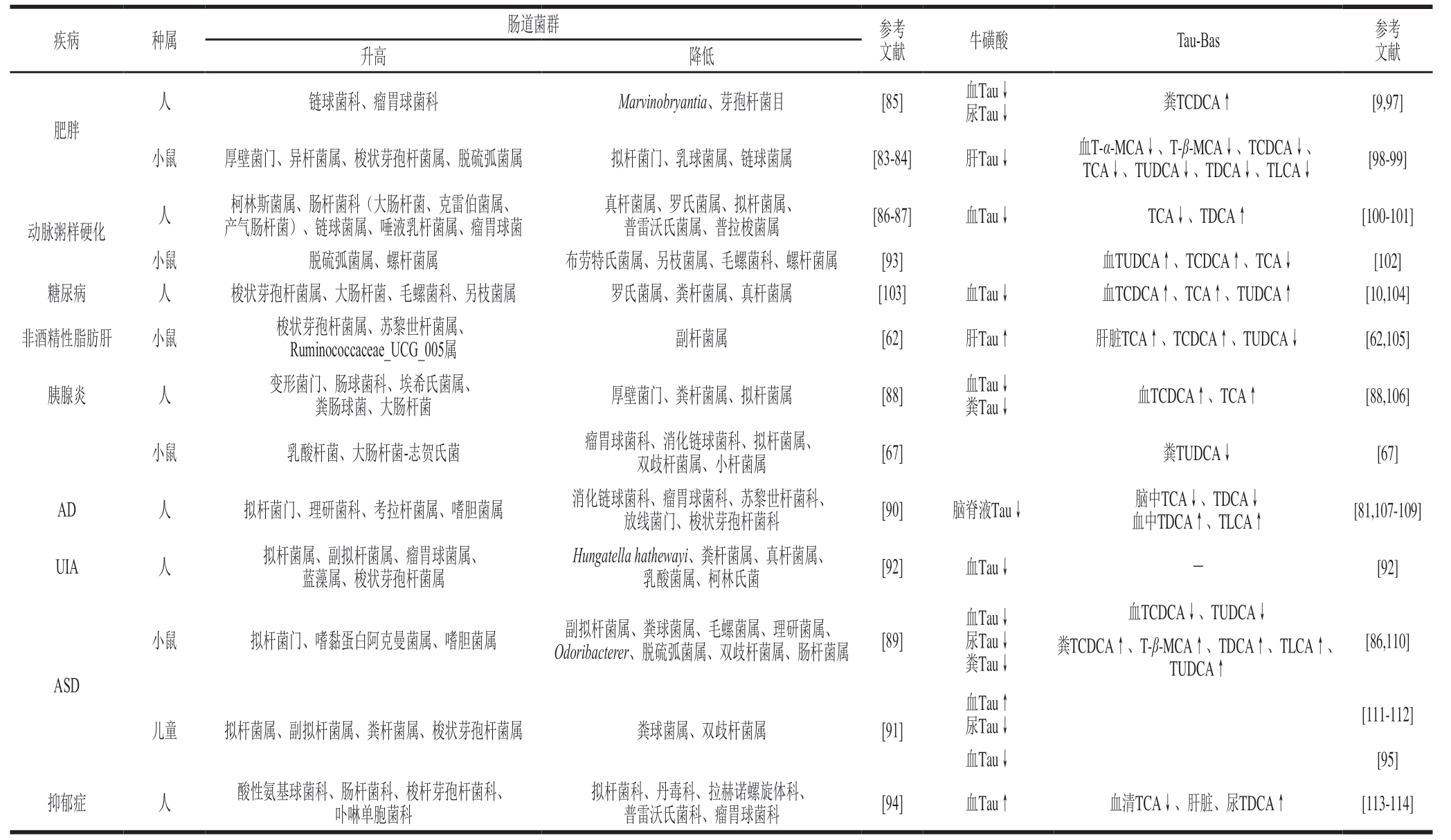
注:↑.升高;↓.降低。
文献牛磺酸Tau-Bas参考文献升高降低肠道菌群疾病种属参考人链球菌科、瘤胃球菌科Marvinobryantia、芽孢杆菌目[85]血Tau↓尿Tau↓粪TCDCA↑[9,97]肥胖小鼠厚壁菌门、异杆菌属、梭状芽孢杆菌属、脱硫弧菌属拟杆菌门、乳球菌属、链球菌属[83-84]肝Tau↓血T-α-MCA↓、T-β-MCA↓、TCDCA↓、TCA↓、TUDCA↓、TDCA↓、TLCA↓[98-99]动脉粥样硬化人柯林斯菌属、肠杆菌科(大肠杆菌、克雷伯菌属、产气肠杆菌)、链球菌属、唾液乳杆菌属、瘤胃球菌真杆菌属、罗氏菌属、拟杆菌属、普雷沃氏菌属、普拉梭菌属[86-87]血Tau↓TCA↓、TDCA↑[100-101]小鼠脱硫弧菌属、螺杆菌属布劳特氏菌属、另枝菌属、毛螺菌科、螺杆菌属[93]血TUDCA↑、TCDCA↑、TCA↓[102]糖尿病人梭状芽孢杆菌属、大肠杆菌、毛螺菌科、另枝菌属罗氏菌属、粪杆菌属、真杆菌属[103]血Tau↓血TCDCA↑、TCA↑、TUDCA↑[10,104]非酒精性脂肪肝小鼠梭状芽孢杆菌属、苏黎世杆菌属、Ruminococcaceae_UCG_005属副杆菌属[62]肝Tau↑肝脏TCA↑、TCDCA↑、TUDCA↓[62,105]胰腺炎人变形菌门、肠球菌科、埃希氏菌属、粪肠球菌、大肠杆菌厚壁菌门、粪杆菌属、拟杆菌属[88]血Tau↓粪Tau↓血TCDCA↑、TCA↑[88,106]小鼠乳酸杆菌、大肠杆菌-志贺氏菌瘤胃球菌科、消化链球菌科、拟杆菌属、双歧杆菌属、小杆菌属[67]粪TUDCA↓[67]AD人拟杆菌门、理研菌科、考拉杆菌属、嗜胆菌属消化链球菌科、瘤胃球菌科、苏黎世杆菌科、放线菌门、梭状芽孢杆菌科[90]脑脊液Tau↓脑中TCA↓、TDCA↓血中TDCA↑、TLCA↑[81,107-109]UIA人拟杆菌属、副拟杆菌属、瘤胃球菌属、蓝藻属、梭状芽孢杆菌属Hungatella hathewayi、粪杆菌属、真杆菌属、乳酸菌属、柯林氏菌[92]血Tau↓-[92]血TCDCA↓、TUDCA↓小鼠拟杆菌门、嗜黏蛋白阿克曼菌属、嗜胆菌属副拟杆菌属、粪球菌属、毛螺菌属、理研菌属、Odoribacterer、脱硫弧菌属、双歧杆菌属、肠杆菌属[89]血Tau↓尿Tau↓粪Tau↓[86,110]粪TCDCA↑、T-β-MCA↑、TDCA↑、TLCA↑、ASD TUDCA↑[111-112]血Tau↓[95]抑郁症人酸性氨基球菌科、肠杆菌科、梭杆芽孢杆菌科、卟啉单胞菌科儿童拟杆菌属、副拟杆菌属、粪杆菌属、梭状芽孢杆菌属粪球菌属、双歧杆菌属[91]血Tau↑尿Tau↓拟杆菌科、丹毒科、拉赫诺螺旋体科、普雷沃氏菌科、瘤胃球菌科[94]血Tau↑血清TCA↓、肝脏、尿TDCA↑[113-114]
此外,大量研究发现,不仅肠道菌群失调和牛磺酸水平改变是疾病发生发展的重要影响因素,Tau-Bas水平的异常也是另一重要的影响因素。如表3所示,这些疾病中,TCA、TCDCA、TUDCA等Tau-Bas常表现为升高,但也存在部分升高和/或部分下降的情况。对AD的研究发现,患者血清中由肠道微生物修饰产生的次级胆汁酸及Tau-Bas(TDCA和TLCA)浓度显著升高[107]。而另一项研究则与之相反,AD患者脑和血浆中的TCA明显降低[108-109]。尽管结果不同,这些研究均显示大脑结构和认知功能损伤与Tau-Bas异常水平之间存在显著关联[107-109]。采用慢性不可预测应激诱导抑郁症小鼠模型,发现拟杆菌门丰度增加,厚壁菌门及乳酸菌丰度降低,同时结肠中TCA水平降低而TUDCA水平升高[113]。由此可见,这些与菌群失调相关的疾病中,牛磺酸的水平普遍呈下降趋势,而Tau-Bas的变化则表现出复杂多样的特征。这可能与Tau-Bas种类繁多以及作为信号分子发挥不同的调节作用有关。多项研究表明,甘氨酸结合型胆汁酸/Tau-Bas比值下降与多种疾病的发生发展存在关联,例如AD、慢性肝病和急性呼吸窘迫综合征,可作为它们的诊断或预期标志物[107,115-116]。这说明研究Tau-Bas的变化应结合整体BAs谱的变化和特定BAs比值从而评估其在疾病发生中的作用。
4.2 补充牛磺酸纠正肠道菌群失调
牛磺酸与肠道微生物间联系密切,如前所述很多疾病同时存在牛磺酸水平下降和肠道菌群失调。直接补充牛磺酸被认为对很多疾病有益,但关于其对肠道微生物影响的研究并不多。如表4所示,不同疾病实验动物经饮水、灌胃、饲料等方式给予牛磺酸,结果发现均可以改善肠道菌群失调。
表4 牛磺酸对肠道微生物的调节
Table 4 Regulation of intestinal microorganisms by taurine
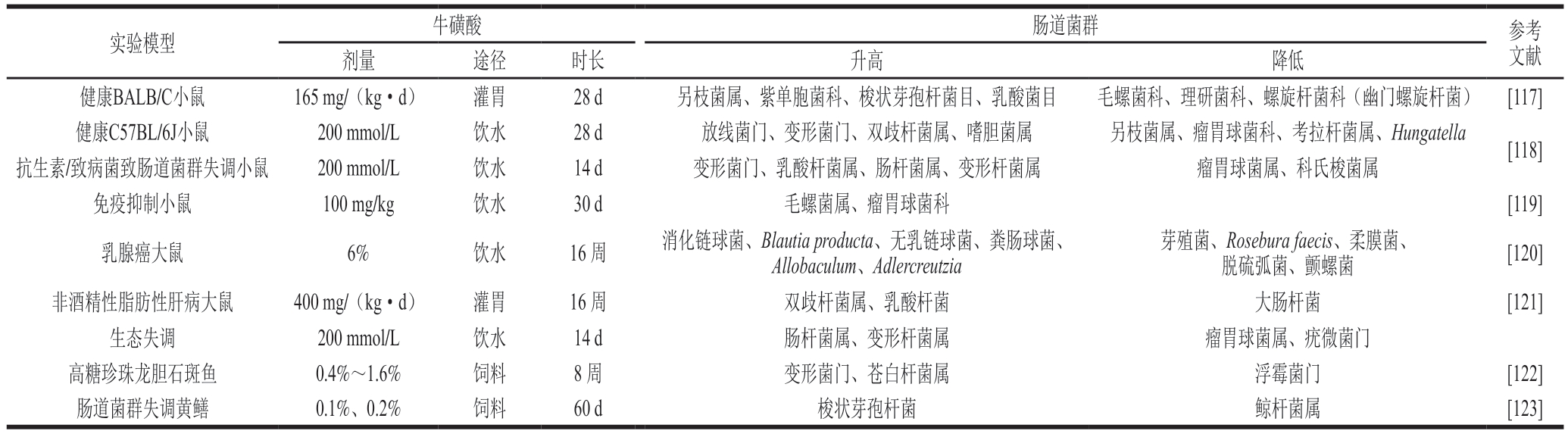
实验模型牛磺酸参考文献剂量途径时长升高降低肠道菌群健康BALB/C小鼠165 mg/(kg·d)灌胃28 d另枝菌属、紫单胞菌科、梭状芽孢杆菌目、乳酸菌目毛螺菌科、理研菌科、螺旋杆菌科(幽门螺旋杆菌)[117]健康C57BL/6J小鼠200 mmol/L饮水28 d放线菌门、变形菌门、双歧杆菌属、嗜胆菌属另枝菌属、瘤胃球菌科、考拉杆菌属、Hungatella[118]抗生素/致病菌致肠道菌群失调小鼠200 mmol/L饮水14 d变形菌门、乳酸杆菌属、肠杆菌属、变形杆菌属瘤胃球菌属、科氏梭菌属免疫抑制小鼠100 mg/kg饮水30 d毛螺菌属、瘤胃球菌科[119]乳腺癌大鼠6%饮水16 周消化链球菌、Blautia producta、无乳链球菌、粪肠球菌、Allobaculum、Adlercreutzia芽殖菌、Rosebura faecis、柔膜菌、脱硫弧菌、颤螺菌[120]非酒精性脂肪性肝病大鼠400 mg/(kg·d)灌胃16 周双歧杆菌属、乳酸杆菌大肠杆菌[121]生态失调200 mmol/L饮水14 d肠杆菌属、变形杆菌属瘤胃球菌属、疣微菌门高糖珍珠龙胆石斑鱼0.4%~1.6%饲料8 周变形菌门、苍白杆菌属浮霉菌门[122]肠道菌群失调黄鳝0.1%、0.2%饲料60 d梭状芽孢杆菌鲸杆菌属[123]
健康小鼠经饮水给予牛磺酸可以显著改变肠道菌群的组成结构,提升菌群多样性,其中双歧杆菌属和嗜胆菌属丰度明显增加[118],可能与它们都是肠道中能够代谢牛磺酸的菌属有关。并且,无论是天然牛磺酸还是合成牛磺酸,均可提高小鼠肠道有益菌乳酸杆菌等菌群丰度,降低变形菌门丰度,特别是抑制有害菌幽门螺杆菌生长[117]。如表4所示,在非酒精脂肪性肝病、高血糖、抗生素处理或致病菌感染致肠道菌群失调等不同疾病模型中,发现牛磺酸的补充能够逆转有益菌乳酸杆菌和双歧杆菌丰度的下降[118,121-122]。牛磺酸对人肠道微生物的直接影响尚鲜见报道。但Sasaki等[70]发现,在模拟人结肠实验中牛磺酸对门和属水平的微生物群组成没有影响,这一结果与以上动物实验的结果并不一致。推测这种差异可能由人类与动物之间的肠道微生物群差异导致。这也可能与牛磺酸的剂量有关。该研究中使用的人类推荐剂量为40 mg/kg(相当于3 g/70 kg成人体质量),将其等效剂量折算到小鼠实验中,该牛磺酸剂量远高于表4所列的实验用量。但有证据显示在改善人肠道菌群时肠道内牛磺酸代谢是上调的[124]。总之,直接补充牛磺酸有助于加强宿主健康,可增加菌群失调宿主肠道中有益菌的丰度,同时降低有害菌丰度。
在小鼠柠檬酸杆菌感染致肠道菌群紊乱的研究中发现,给予牛磺酸可以显著降低致病菌的载量,并且能够抑制感染小鼠的体质量下降和改善肠道微生物多样性的降低,可见牛磺酸增强了宿主对致病菌的定植抗性[118]。而Stacy等[125]发现短暂感染食源性病原体鼠疫减毒菌株(DyopM)后可改变肠道菌群,并可增强对致病菌的定植抗性。进一步研究揭示其机制,短暂感染后肠道内牛磺酸水平升高,使得牛磺酸代谢菌群扩张,可将牛磺酸代谢为硫化物,这一硫化物通过阻断病原体有氧呼吸而增强了宿主对致病菌定植抗力。随后进一步实验证实,单独补充牛磺酸或移植牛磺酸处理过的粪便微生物都会复制这种微生物群功能并增强定植抗性[125]。总之,牛磺酸可被视为调整肠道微生物群、提高定植抗性的营养物质。
牛磺酸改善肠道菌群影响机体健康的机制可能是改善菌群代谢物。胆汁酸和LPS是常见的两种肠道菌群代谢物。补充牛磺酸可以显著增加肠道TUDCA、TLCA、THDCA、TDCA、TCA和T-β-MCA等Tau-Bas含量[118],Tau-Bas不仅促进有益菌丰度增加[65-68],也可以调节NK细胞而增强免疫力[52]。LPS是大肠杆菌等革兰氏阴性菌细胞膜上内毒素的主要成分,是炎症反应的有效激活剂,LPS不仅能够直接引起急性肠黏膜炎,还可经肠壁吸收进入循环而影响全身,导致肝脂肪变性及纤维化[126]。研究发现,牛磺酸降低了大肠杆菌等革兰氏阴性菌的丰度和LPS浓度[117,121]。Zhu Wenhua等[127]也发现补充牛磺酸治疗HFD诱导的脂肪肝大鼠,降低了血清LPS水平,缓解了肝细胞损伤和氧化应激。可见牛磺酸不仅直接降低释放LPS的菌群丰度,还可促进生成Tau-Bas而提高相应有益菌的丰度,表现出有益于重塑健康的肠道菌群作用。
另一方面牛磺酸还可增强机体免疫功能。在一项免疫抑制小鼠实验中,补充牛磺酸后肠道菌群明显改善,并且免疫细胞数量以及T细胞(CD3+)和B细胞(CD19+)亚群显著升高[119]。研究表明,T细胞膜上TauT敲除会导致T细胞激活功能受损,证实牛磺酸对T细胞存活及其介导的免疫功能至关重要[128]。给予抗生素不仅抑制小鼠肠道菌群丰度,还造成结肠组织细胞因子白细胞介素(interleukin,IL)-6、IL-17和肿瘤坏死因子-α(tumor necrosis factor-α,TNF-α)含量明显下降,而牛磺酸补充消除了抗生素对宿主这些免疫分子的负面影响[118]。一项离体细胞实验结果证实,牛磺酸可以抑制TNF-α诱导的人肠上皮Caco-2细胞分泌IL-8,保护肠上皮细胞[129]。NLRP6炎症小体在肠道微生物稳态调节中发挥关键作用。牛磺酸激活NLRP6炎症小体生成IL-18的能力表现出显著的剂量依赖性[130]。使用IL-23抑制剂治疗银屑病患者,发现其肠道细菌组成改变,并且牛磺酸代谢也显著增加[124]。可见,牛磺酸不仅对T细胞的存活和功能至关重要,还可以提高包括T细胞在内的免疫细胞数量。而细胞因子作为免疫细胞功能的重要执行分子,牛磺酸表现出对免疫细胞因子分泌的促进作用。总之,牛磺酸能够提高机体的免疫功能。
综上所述,牛磺酸可刺激牛磺酸代谢菌群的丰度扩大,同时通过特定肠道菌群促进次级胆汁酸生成、有益菌丰度增加,以及直接提高机体免疫力从而增强机体抵抗有害菌定植的能力(图3),这些都有助于牛磺酸发挥肠道菌群稳态的重塑作用。
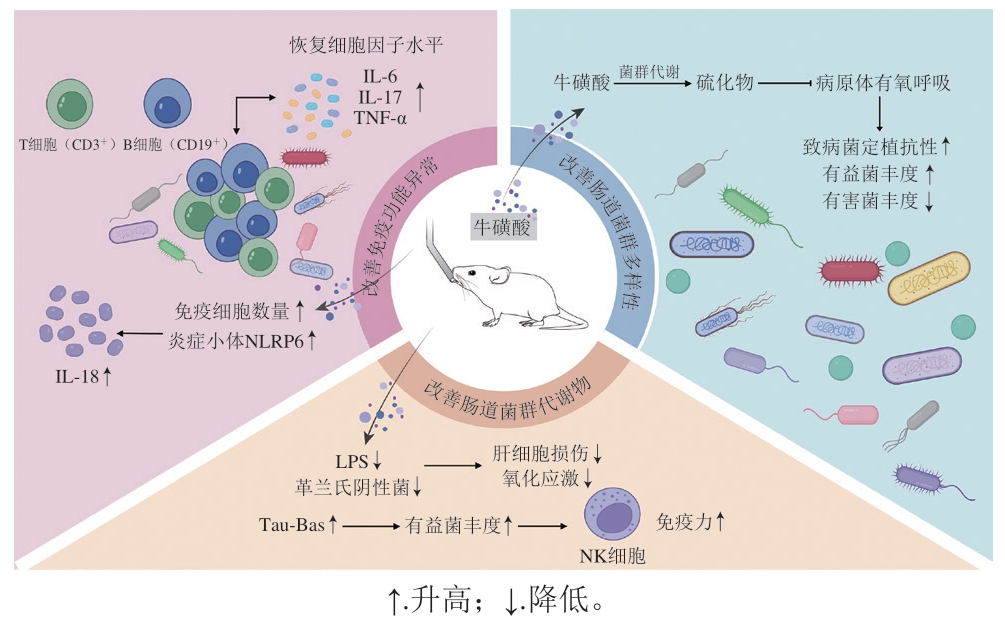
图3 牛磺酸对疾病所致肠道微生物紊乱的调节作用
Fig. 3 Regulatory effect of taurine on intestinal microflora disorders caused by diseases
5 结 语
牛磺酸和胆汁酸代谢紧密相关,是肠道微生物的两种重要代谢产物。牛磺酸/胆汁酸与肠道微生物之间相互作用、相互影响,共同构成了一个功能调节轴,对机体的生理过程有重要影响。牛磺酸在促进肠道菌群健康和调节肠道菌群稳态重塑方面具有很好的潜力。因此,进一步深入研究牛磺酸对人肠道菌群的直接影响,以及调节肠道菌群的分子机制和靶点,有助于未来将牛磺酸加入常用的肠道微生物调节方法中,比如联合开发牛磺酸和/或益生菌,作为预防和改善肠道菌群失调相关疾病的备选方案。
[1] LIN S L, WANG Z Y, LAM K L, et al. Role of intestinal microecology in the regulation of energy metabolism by dietary polyphenols and their metabolites[J]. Food & Nutrition Research, 2019, 63: 63.DOI:10.29219/fnr.v63.1518.
[2] LOZUPONE C A, STOMBAUGH J I, GORDON J I, et al. Diversity,stability and resilience of the human gut microbiota[J]. Nature, 2012,489(7415): 220-230. DOI:10.1038/nature11550.
[3] 王晨, 钟赛意, 邹宇晓. 膳食纤维经肠道微生态途径调节脂质代谢作用的研究进展[J]. 食品科学, 2019, 40(3): 338-347. DOI:10.7506/spkx1002-6630-20171016-114.
[4] MINCIC A M, ANTAL M, FILIP L, et al. Modulation of gut microbiome in the treatment of neurodegenerative diseases: a systematic review[J]. Clinical Nutrition, 2024, 43(7): 1832-1849.DOI:10.1016/j.clnu.2024.05.036.
[5] ZHANG C H, ZHANG M H, WANG S Y, et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice[J]. The ISME Journal, 2010, 4(2): 232-241. DOI:10.1038/ismej.2009.112.
[6] VIJAY A, ASTBURY S, LE ROY C, et al. The prebiotic effects of omega-3 fatty acid supplementation: a six-week randomised intervention trial[J]. Gut Microbes, 2021, 13(1): 1-11. DOI:10.1080/19490976.2020.1863133.
[7] SCHULLER-LEVIS G B, PARK E. Taurine: new implications for an old amino acid[J]. FEMS Microbiology Letters, 2003, 226(2): 195-202. DOI:10.1016/S0378-1097(03)00611-6.
[8] SEIDEL U, HUEBBE P, RIMBACH G. Taurine: a regulator of cellular redox homeostasis and skeletal muscle function[J]. Molecular Nutrition & Food Research, 2019, 63(16): e1800569. DOI:10.1002/mnfr.201800569.
[9] ROSA F T, FREITAS E C, DEMINICE R, et al. Oxidative stress and inflammation in obesity after taurine supplementation: a double-blind,placebo-controlled study[J]. European Journal of Nutrition, 2014,53(3): 823-830. DOI:10.1007/s00394-013-0586-7.
[10] CALVANI R, RODRIGUEZ-MAÑAS L, PICCA A, et al. Identification of a circulating amino acid signature in frail older persons with type 2 diabetes mellitus: results from the metabofrail study[J]. Nutrients,2020, 12(1): 199. DOI:10.3390/nu12010199.
[11] YAMORI Y, TAGUCHI T, MORI H, et al. Low cardiovascular risks in the middle aged males and females excreting greater 24-hour urinary taurine and magnesium in 41 WHO-CARDIAC study populations in the world[J]. Journal of Biomedical Science, 2010, 17(Suppl 1): S21.DOI:10.1186/1423-0127-17-S1-S21.
[12] SAGARA M, MURAKAMI S, MIZUSHIMA S, et al. Taurine in 24-h urine samples is inversely related to cardiovascular risks of middle aged subjects in 50 populations of the world[J]. Advances in Experimental Medicine and Biology, 2015, 803: 623-636.DOI:10.1007/978-3-319-15126-7_50.
[13] ISHIKAWA M, ARAI S, TAKANO M, et al. Taurine’s health influence on Japanese high school girls[J]. Journal of Biomedical Science, 2010,17(Suppl 1): S47. DOI:10.1186/1423-0127-17-S1-S47.
[14] CLEMENTE-SUÁREZ V J, RAMÍREZ-GOERKE M I, REDONDOFLÓREZ L, et al. The impact of anorexia nervosa and the basis for non-pharmacological interventions[J]. Nutrients, 2023, 15(11): 2594.DOI:10.3390/nu15112594.
[15] MOHR A, JÄGER R, CARPENTER K, et al. The athletic gut microbiota[J]. Journal of the International Society of Sports Nutrition,2020, 17(1): 24. DOI:10.1186/s12970-020-00353-w.
[16] CHANG Y L, OU Q L, ZHOU X H, et al. Global research trends and focus on the link between colorectal cancer and gut flora: a bibliometric analysis from 2001 to 2021[J]. Frontiers in Microbiology,2023, 14: 1182006. DOI:10.3389/fmicb.2023.1182006.
[17] 刘昭志, 彭莉. 运动、肠道菌群和2型糖尿病关系的研究进展[J]. 微生物学报, 2025, 65(1): 1-16. DOI:10.13343/j.cnki.wsxb.20240485.
[18] HUANG H L, JIA C X, CHEN X Y, et al. Progress in research on the effects of quinoa (Chenopodium quinoa) bioactive compounds and products on intestinal flora[J]. Frontiers in Nutrition, 2024, 11:1308384. DOI:10.3389/fnut.2024.1308384.
[19] KRAUTKRAMER K A, FAN J, BÄCKHED F. Gut microbial metabolites as multi-kingdom intermediates[J]. Nature Reviews.Microbiology, 2021, 19(2): 77-94. DOI:10.1038/s41579-020-0438-4.
[20] JACOBSEN J G, SMITH L H. Biochemistry and physiology of taurine and taurine derivatives[J]. Physiological Reviews, 1968, 48(2): 424-511. DOI:10.1152/physrev.1968.48.2.424.
[21] EDGAR S E, KIRK C A, ROGERS Q R, et al. Taurine status in cats is not maintained by dietary cysteinesulfinic acid[J]. The Journal of Nutrition, 1998, 128(4): 751-757. DOI:10.1093/jn/128.4.751.
[22] LAMBERT I H, KRISTENSEN D M, HOLM J B, et al. Physiological role of taurine: from organism to organelle[J]. Acta Physiologica,2015, 213(1): 191-212. DOI:10.1111/apha.12365.
[23] CHESNEY R W, HELMS R A, CHRISTENSEN M, et al. The role of taurine in infant nutrition[J]. Advances in Experimental Medicine and Biology, 1998, 442: 463-476. DOI:10.1007/978-1-4899-0117-0_56.
[24] WU G Y. Important roles of dietary taurine, creatine, carnosine,anserine and 4-hydroxyproline in human nutrition and health[J]. Amino Acids, 2020, 52(3): 329-360. DOI:10.1007/s00726-020-02823-6.
[25] SANTULLI G, KANSAKAR U, VARZIDEH F, et al. Functional role of taurine in aging and cardiovascular health: an updated overview[J].Nutrients, 2023, 15(19): 4236. DOI:10.3390/nu15194236.
[26] ZHAO X, JIA J, LIN Y. Taurine content in Chinese food and daily intake of Chinese men[J]. Advances in Experimental Medicine and Biology, 1998, 442: 501-505. DOI:10.1007/978-1-4899-0117-0_60.
[27] CHEN X M, YOU J M, SUO Y R, et al. Sensitive determination of taurine, γ-aminobutyric acid and ornithine in wolfberry fruit and cortex lycii by HPLC with fluorescence detection and online mass spectrometry identification[J]. Journal of Chromatographic Science,2015, 53(4): 492-497. DOI:10.1093/chromsci/bmu072.
[28] ZHAO Z, SATSU H, FUJISAWA M, et al. Attenuation by dietary taurine of dextran sulfate sodium-induced colitis in mice and of THP-1-induced damage to intestinal Caco-2 cell monolayers[J]. Amino Acids, 2008, 35(1): 217-224. DOI:10.1007/s00726-007-0562-8.
[29] BALIOU S, KYRIAKOPOULOS A M, GOULIELMAKI M, et al.Significance of taurine transporter (TauT) in homeostasis and its layers of regulation[J]. Molecular Medicine Reports, 2020, 22(3): 2163-2173.DOI:10.3892/mmr.2020.11321.
[30] HELLER-STILB B, VAN ROEYEN C, RASCHER K, et al. Disruption of the taurine transporter gene (taut) leads to retinal degeneration in mice[J]. The FASEB Journal, 2002, 16(2): 231-233. DOI:10.1096/fj.01-0691fje.
[31] WARSKULAT U, BORSCH E, REINEHR R, et al. Taurine deficiency and apoptosis: findings from the taurine transporter knockout mouse[J]. Archives of Biochemistry and Biophysics, 2007, 462(2):202-209. DOI:10.1016/j.abb.2007.03.022.
[32] HAN X B, CHESNEY R W. Knockdown of TauT expression impairs human embryonic kidney 293 cell development[J]. Advances in Experimental Medicine and Biology, 2013, 776: 307-320.DOI:10.1007/978-1-4614-6093-0_28.
[33] DONEPUDI A C, BOEHME S, LI F, et al. G-protein-coupled bile acid receptor plays a key role in bile acid metabolism and fastinginduced hepatic steatosis in mice[J]. Hepatology, 2017, 65(3): 813-827. DOI:10.1002/hep.28707.
[34] SJOVALL J. Dietary glycine and taurine on bile acid conjugation in man; bile acids and steroids 75[J]. Proceedings of the Society for Experimental Biology and Medicine, 1959, 100(4): 676-678.DOI:10.3181/00379727-100-24741.
[35] FIORUCCI S, CARINO A, BALDONI M, et al. Bile acid signaling in inflammatory bowel diseases[J]. Digestive Diseases and Sciences,2021, 66(3): 674-693. DOI:10.1007/s10620-020-06715-3.
[36] JIA W, XIE G X, JIA W P. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis[J]. Nature Reviews Gastroenterology & Hepatology, 2018, 15(2): 111-128. DOI:10.1038/nrgastro.2017.119.
[37] KIRIYAMA Y, NOCHI H. The biosynthesis, signaling, and neurological functions of bile acids[J]. Biomolecules, 2019, 9(6): 232.DOI:10.3390/biom9060232.
[38] STURMAN J A, HEPNER G W, HOFMANN A F, et al. Metabolism of [35S] taurine in man[J]. The Journal of Nutrition, 1975, 105(9):1206-1214. DOI:10.1093/jn/105.9.1206.
[39] FUCHS C D, TRAUNER M. Role of bile acids and their receptors in gastrointestinal and hepatic pathophysiology[J]. Nature Reviews Gastroenterology & Hepatology, 2022, 19(7): 432-450. DOI:10.1038/s41575-021-00566-7.
[40] RAMÍREZ-PÉREZ O, CRUZ-RAMÓN V, CHINCHILLA-LÓPEZ P,et al. The role of the gut microbiota in bile acid metabolism[J]. Annals of Hepatology, 2017, 16: S21-S26. DOI:10.5604/01.3001.0010.5672.
[41] CHEN W, GUO J X, ZHANG Y Z, et al. The beneficial effects of taurine in preventing metabolic syndrome[J]. Food & Function, 2016,7(4): 1849-1863. DOI:10.1039/C5FO01295C.
[42] BROWN A W W, WILSON R B. Clostridium difficile colitis and zoonotic origins: a narrative review[J]. Gastroenterology Report, 2018,6(3): 157-166. DOI:10.1093/gastro/goy016.
[43] CALICETI C, PUNZO A, SILLA A, et al. New insights into bile acids related signaling pathways in the onset of colorectal cancer[J].Nutrients, 2022, 14(14): 2964. DOI:10.3390/nu14142964.
[44] 李楚荞, 刘天宇, 宋雪例, 等. 胆汁酸-肠道菌群相互作用与结直肠癌发生发展的研究进展[J]. 肿瘤药学, 2019, 9(5): 710-715.DOI:10.3969/j.issn.2095-1264.2019.05.02.
[45] KISIELA M, SKARKA A, EBERT B, et al. Hydroxysteroid dehydrogenases (HSDs) in bacteria: a bioinformatic perspective[J].The Journal of Steroid Biochemistry and Molecular Biology, 2012,129(1/2): 31-46. DOI:10.1016/j.jsbmb.2011.08.002.
[46] KENDALL M M, SPERANDIO V. Gut microbes regroup to aid defence after infection[J]. Nature, 2021, 592(7852): 29-31.DOI:10.1038/d41586-021-00642-7.
[47] SONG Z W, CAI Y Y, LAO X Z, et al. Taxonomic profiling and populational patterns of bacterial bile salt hydrolase (BSH) genes based on worldwide human gut microbiome[J]. Microbiome, 2019,7(1): 9. DOI:10.1186/s40168-019-0628-3.
[48] SUN L L, ZHANG Y, CAI J, et al. Bile salt hydrolase in nonenterotoxigenic Bacteroides potentiates colorectal cancer[J]. Nature Communications, 2023, 14(1): 755. DOI:10.1038/s41467-023-36089-9.
[49] HUANG F J, ZHENG X J, MA X H, et al. Theabrownin from Pu-erh tea attenuates hypercholesterolemia via modulation of gut microbiota and bile acid metabolism[J]. Nature Communications, 2019, 10(1):4971. DOI:10.1038/s41467-019-12896-x.
[50] SWANN J R, WANT E J, GEIER F M, et al. Systemic gut microbial modulation of bile acid metabolism in host tissue compartments[J].Proceedings of the National Academy of Sciences of the United States of America, 2011, 108(Suppl 1): 4523-4530. DOI:10.1073/pnas.1006734107.
[51] CLAUS S P, TSANG T M, WANG Y L, et al. Systemic multicompartmental effects of the gut microbiome on mouse metabolic phenotypes[J]. Molecular Systems Biology, 2008, 4: 219.DOI:10.1038/msb.2008.56.
[52] MA C, HAN M J, HEINRICH B, et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells[J]. Science,2018, 360(6391): eaan5931. DOI:10.1126/science.aan5931.
[53] CHEN W J, ZHOU S N, XIAO J C, et al. Farnesoid X receptor is inhibited after ileum transposition in diabetic rats: its hypoglycemic effect[J]. International Journal of Medical Sciences, 2023, 20(5): 595-605. DOI:10.7150/ijms.80563.
[54] BAARS A, OOSTING A, LOHUIS M, et al. Sex differences in lipid metabolism are affected by presence of the gut microbiota[J]. Scientific Reports, 2018, 8(1): 13426. DOI:10.1038/s41598-018-31695-w.
[55] SARNI R O S, KOCHI C, SUANO-SOUZA F I. Childhood obesity:an ecological perspective[J]. Jornal de Pediatria, 2022, 98(Suppl 1):S38-S46. DOI:10.1016/j.jped.2021.10.002.
[56] DUBOC H, TACHÉ Y, HOFMANN A F. The bile acid TGR5 membrane receptor: from basic research to clinical application[J].Digestive and Liver Disease, 2014, 46(4): 302-312. DOI:10.1016/j.dld.2013.10.021.
[57] GUO C, CHEN W D, WANG Y D. TGR5, not only a metabolic regulator[J]. Frontiers in Physiology, 2016, 7: 646. DOI:10.3389/fphys.2016.00646.
[58] KONG B, HUANG J S, ZHU Y, et al. Fibroblast growth factor 15 deficiency impairs liver regeneration in mice[J]. American Journal of Physiology Gastrointestinal and Liver Physiology, 2014, 306(10):G893-G902. DOI:10.1152/ajpgi.00337.2013.
[59] MIYAZAKI T. Identification of a novel enzyme and the regulation of key enzymes in mammalian taurine synthesis[J]. Journal of Pharmacological Sciences, 2024, 154(1): 9-17. DOI:10.1016/j.jphs.2023.11.003.
[60] SAYIN S I, WAHLSTRÖM A, FELIN J, et al. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist[J]. Cell Metabolism, 2013,17(2): 225-235. DOI:10.1016/j.cmet.2013.01.003.
[61] MÜNZKER J, HAASE N, TILL A, et al. Functional changes of the gastric bypass microbiota reactivate thermogenic adipose tissue and systemic glucose control via intestinal FXR-TGR5 crosstalk in dietinduced obesity[J]. Microbiome, 2022, 10(1): 96. DOI:10.1186/s40168-022-01264-5.
[62] LEI Y Y, TANG L, CHEN Q, et al. Disulfiram ameliorates nonalcoholic steatohepatitis by modulating the gut microbiota and bile acid metabolism[J]. Nature Communications, 2022, 13(1): 6862.DOI:10.1038/s41467-022-34671-1.
[63] LIU Y H, CHEN K F, LI F Y, et al. Probiotic Lactobacillus rhamnosus GG prevents liver fibrosis through inhibiting hepatic bile acid synthesis and enhancing bile acid excretion in mice[J]. Hepatology, 2020, 71(6):2050-2066. DOI:10.1002/hep.30975.
[64] LI F, JIANG C T, KRAUSZ K W, et al. Microbiome remodelling leads to inhibition of intestinal farnesoid X receptor signalling and decreased obesity[J]. Nature Communications, 2013, 4: 2384. DOI:10.1038/ncomms3384.
[65] WANG W J, ZHAO J F, GUI W F, et al. Tauroursodeoxycholic acid inhibits intestinal inflammation and barrier disruption in mice with non-alcoholic fatty liver disease[J]. British Journal of Pharmacology,2018, 175(3): 469-484. DOI:10.1111/bph.14095.
[66] SONG M, ZHANG F L, FU Y M, et al. Tauroursodeoxycholic acid (TUDCA) improves intestinal barrier function associated with TGR5-MLCK pathway and the alteration of serum metabolites and gut bacteria in weaned piglets[J]. Journal of Animal Science and Biotechnology, 2022, 13(1): 73. DOI:10.1186/s40104-022-00713-3.
[67] WAN Y D, ZHU R X, PAN X T, et al. Bile acid supplementation improves murine pancreatitis in association with the gut microbiota[J]. Frontiers in Physiology, 2020, 11: 650. DOI:10.3389/fphys.2020.00650.
[68] XU J, XIE S W, CHI S Y, et al. Protective effects of taurocholic acid on excessive hepatic lipid accumulation via regulation of bile acid metabolism in grouper[J]. Food & Function, 2022, 13(5): 3050-3062.DOI:10.1039/d1fo04085e.
[69] VAN DER PLOEG J R, WEISS M A, SALLER E, et al. Identification of sulfate starvation-regulated genes in Escherichia coli: a gene cluster involved in the utilization of taurine as a sulfur source[J].Journal of Bacteriology, 1996, 178(18): 5438-5446. DOI:10.1128/jb.178.18.5438-5446.1996.
[70] SASAKI K, SASAKI D, OKAI N, et al. Taurine does not affect the composition, diversity, or metabolism of human colonic microbiota simulated in a single-batch fermentation system[J]. PLoS ONE, 2017,12(7): e0180991. DOI:10.1371/journal.pone.0180991.
[71] HUXTABLE R J. Physiological actions of taurine[J]. Physiological Reviews, 1992, 72(1): 101-163. DOI:10.1152/physrev.1992.72.1.101.
[72] JAVAUX C, JORIS B, DE WITTE P. Functional characteristics of TauA binding protein from TauABC Escherichia coli system[J]. The Protein Journal, 2007, 26(4): 231-238. DOI:10.1007/s10930-006-9064-x.
[73] EICHHORN E, VAN DER PLOEG J R, LEISINGER T. Deletion analysis of the Escherichia coli taurine and alkanesulfonate transport systems[J]. Journal of Bacteriology, 2000, 182(10): 2687-2695.DOI:10.1128/JB.182.10.2687-2695.2000.
[74] QUADRONI M, JAMES P, DAINESE-HATT P, et al. Proteome mapping, mass spectrometric sequencing and reverse transcription-PCR for characterization of the sulfate starvation-induced response in Pseudomonas aeruginosa PAO1[J]. European Journal of Biochemistry,1999, 266(3): 986-996. DOI:10.1046/j.1432-1327.1999.00941.x.
[75] VAN DER PLOEG J R, CUMMINGS N J, LEISINGER T,et al. Bacillus subtilis genes for the utilization of sulfur from aliphatic sulfonates[J]. Microbiology, 1998, 144: 2555-2561.DOI:10.1099/00221287-144-9-2555.
[76] MASEPOHL B, FÜHRER F, KLIPP W. Genetic analysis of a Rhodobacter capsulatus gene region involved in utilization of taurine as a sulfur source[J]. FEMS Microbiology Letters, 2001, 205(1): 105-111. DOI:10.1016/S0378-1097(01)00447-5.
[77] WIETHAUS J, SCHUBERT B, PFÄNDER Y, et al. The GntR-like regulator TauR activates expression of taurine utilization genes in Rhodobacter capsulatus[J]. Journal of Bacteriology, 2008, 190(2):487-493. DOI:10.1128/JB.01510-07.
[78] DENGER K, SMITS T H M, COOK A M. Genome-enabled analysis of the utilization of taurine as sole source of carbon or of nitrogen by Rhodobacter sphaeroides 2.4.1[J]. Microbiology, 2006, 152: 3197-3206. DOI:10.1099/mic.0.29195-0.
[79] HAANGE S B, GROEGER N, FROMENT J, et al. Multiplexed quantitative assessment of the fate of taurine and sulfoquinovose in the intestinal microbiome[J]. Metabolites, 2020, 10(11): 430.DOI:10.3390/metabo10110430.
[80] LI M Y, WEI Y F, YIN J Y, et al. Biochemical and structural investigation of taurine: 2-oxoglutarate aminotransferase from Bifidobacterium kashiwanohense[J]. Biochemical Journal, 2019,476(11): 1605-1619. DOI:10.1042/BCJ20190206.
[81] ALOM J, MAHY J N, BRANDI N, et al. Cerebrospinal fluid taurine in Alzheimer’s disease[J]. Annals of Neurology, 1991, 30(5): 735.DOI:10.1002/ana.410300518.
[82] DU Y, GAO X R, PENG L, et al. Crosstalk between the microbiotagut-brain axis and depression[J]. Heliyon, 2020, 6(6): e04097.DOI:10.1016/j.heliyon.2020.e04097.
[83] LEY R E, BÄCKHED F, TURNBAUGH P, et al. Obesity alters gut microbial ecology[J]. Proceedings of the National Academy of Sciences of the United States of America, 2005, 102(31): 11070-11075. DOI:10.1073/pnas.0504978102.
[84] ZHANG H X, CHEN S Q, YANG L, et al. Distinct gut microbiota and arachidonic acid metabolism in obesity-prone and obesityresistant mice with a high-fat diet[J]. Nutrients, 2024, 16(11): 1579.DOI:10.3390/nu16111579.
[85] SHANTARAM D, HOYD R, BLASZCZAK A M, et al. Obesityassociated microbiomes instigate visceral adipose tissue inflammation by recruitment of distinct neutrophils[J]. Nature Communications,2024, 15(1): 5434. DOI:10.1038/s41467-024-48935-5.
[86] JIE Z Y, XIA H H, ZHONG S L, et al. The gut microbiome in atherosclerotic cardiovascular disease[J]. Nature Communications,2017, 8(1): 845. DOI:10.1038/s41467-017-00900-1.
[87] KARLSSON F H, FÅK F, NOOKAEW I, et al. Symptomatic atherosclerosis is associated with an altered gut metagenome[J].Nature Communications, 2012, 3: 1245. DOI:10.1038/ncomms2266.
[88] LI G Q, LIU L W, LU T Q, et al. Gut microbiota aggravates neutrophil extracellular traps-induced pancreatic injury in hypertriglyceridemic pancreatitis[J]. Nature Communications, 2023, 14(1): 6179.DOI:10.1038/s41467-023-41950-y.
[89] GOLUBEVA A V, JOYCE S A, MOLONEY G, et al. Microbiotarelated changes in bile acid & tryptophan metabolism are associated with gastrointestinal dysfunction in a mouse model of autism[J].EBioMedicine, 2017, 24: 166-178. DOI:10.1016/j.ebiom.2017.09.020.
[90] VOGT N M, KERBY R L, DILL-MCFARLAND K A, et al. Gut microbiome alterations in Alzheimer’s disease[J]. Scientific Reports,2017, 7(1): 13537. DOI:10.1038/s41598-017-13601-y.
[91] IGLESIAS-VÁZQUEZ L, VAN GINKEL RIBA G, ARIJA V, et al.Composition of gut microbiota in children with autism spectrum disorder: a systematic review and meta-analysis[J]. Nutrients, 2020,12(3): 792. DOI:10.3390/nu12030792.
[92] LI H, XU H C, LI Y X, et al. Alterations of gut microbiota contribute to the progression of unruptured intracranial aneurysms[J]. Nature Communications, 2020, 11(1): 3218. DOI:10.1038/s41467-020-16990-3.
[93] LI J Y, YANG S Q, LIU D, et al. Neoagarotetraose alleviates atherosclerosis via modulating cholesterol and bile acid metabolism in ApoE-/- mice[J]. Nutrients, 2024, 16(10): 1502. DOI:10.3390/nu16101502.
[94] JIANG H Y, LING Z X, ZHANG Y H, et al. Altered fecal microbiota composition in patients with major depressive disorder[J]. Brain,Behavior, and Immunity, 2015, 48: 186-194. DOI:10.1016/j.bbi.2015.03.016.
[95] PARK E, COHEN I, GONZALEZ M, et al. Is taurine a biomarker in autistic spectrum disorder?[J]. Advances in Experimental Medicine and Biology, 2017, 975: 3-16. DOI:10.1007/978-94-024-1079-2_1.
[96] SHARON G, CRUZ N J, KANG D W, et al. Human gut microbiota from autism spectrum disorder promote behavioral symptoms in mice[J]. Cell, 2019, 177(6): 1600-1618.e17. DOI:10.1016/j.cell.2019.05.004.
[97] 洪泽慧, 罗若城, 董科, 等. 四川省藏汉肥胖与正常人群肠道菌群及粪便胆汁酸差异研究[J]. 现代预防医学, 2022, 49(23): 4351-4357.DOI:10.20043/j.cnki.MPM.202206433.
[98] DING L L, YANG Q L, ZHANG E Y, et al. Notoginsenoside Ft1 acts as a TGR5 agonist but FXR antagonist to alleviate high fat dietinduced obesity and insulin resistance in mice[J]. Acta Pharmaceutica Sinica B, 2021, 11(6): 1541-1554. DOI:10.1016/j.apsb.2021.03.038.
[99] ZHOU W, DU Z. Oleuropein mitigates non-alcoholic fatty liver disease(NAFLD) and modulates liver metabolites in high-fat diet-induced obese mice via activating PPARα[J]. Journal of the Science of Food and Agriculture, 2024, 104(14): 8634-8645. DOI:10.1002/jsfa.13691.
[100] LIU H H, CHEN X, HU X M, et al. Alterations in the gut microbiome and metabolism with coronary artery disease severity[J]. Microbiome,2019, 7(1): 68. DOI:10.1186/s40168-019-0683-9.
[101] SU J R, ZHAO Q, ZHAO A H, et al. Serum metabolic signatures of subclinical atherosclerosis in patients with type 2 diabetes mellitus:a preliminary study[J]. Acta Diabetologica, 2021, 58(9): 1217-1224.DOI:10.1007/s00592-021-01717-7.
[102] XU Y Y, LI Y Y, JADHAV K, et al. Hepatocyte ATF3 protects against atherosclerosis by regulating HDL and bile acid metabolism[J]. Nature Metabolism, 2021, 3(1): 59-74. DOI:10.1038/s42255-020-00331-1.
[103] QIN J J, LI Y R, CAI Z M, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes[J]. Nature, 2012, 490(7418):55-60. DOI:10.1038/nature11450.
[104] WEWALKA M, PATTI M E, BARBATO C, et al. Fasting serum taurine-conjugated bile acids are elevated in type 2 diabetes and do not change with intensification of insulin[J]. The Journal of Clinical Endocrinology and Metabolism, 2014, 99(4): 1442-1451.DOI:10.1210/jc.2013-3367.
[105] LAKE A D, NOVAK P, SHIPKOVA P, et al. Decreased hepatotoxic bile acid composition and altered synthesis in progressive human nonalcoholic fatty liver disease[J]. Toxicology and Applied Pharmacology, 2013, 268(2): 132-140. DOI:10.1016/j.taap.2013.01.022.
[106] ZHU Q T, YUAN C C, DONG X W, et al. Bile acid metabolomics identifies chenodeoxycholic acid as a therapeutic agent for pancreatic necrosis[J]. Cell Reports Medicine, 2023, 4(12): 101304.DOI:10.1016/j.xcrm.2023.101304.
[107] MAHMOUDIANDEHKORDI S, ARNOLD M, NHO K, et al. Altered bile acid profile associates with cognitive impairment in Alzheimer’s disease: an emerging role for gut microbiome[J]. Alzheimer’s &Dementia, 2019, 15(1): 76-92. DOI:10.1016/j.jalz.2018.07.217.
[108] NHO K, KUEIDER-PAISLEY A, MAHMOUDIANDEHKORDI S,et al. Altered bile acid profile in mild cognitive impairment and Alzheimer’s disease: relationship to neuroimaging and CSF biomarkers[J]. Alzheimer’s & Dementia, 2019, 15(2): 232-244.DOI:10.1016/j.jalz.2018.08.012.
[109] PAN X B, ELLIOTT C T, MCGUINNESS B, et al. Metabolomic profiling of bile acids in clinical and experimental samples of Alzheimer’s disease[J]. Metabolites, 2017, 7(2): 28. DOI:10.3390/metabo7020028.
[110] WU J F, HU Q Y, RAO X P, et al. Gut microbiome and metabolic profiles of mouse model for MeCP2 duplication syndrome[J].Brain Research Bulletin, 2024, 206: 110862. DOI:10.1016/j.brainresbull.2023.110862.
[111] XU X J, CAI X E, MENG F C, et al. Comparison of the metabolic profiles in the plasma and urine samples between autistic and typically developing boys: a preliminary study[J]. Frontiers in Psychiatry, 2021,12: 657105. DOI:10.3389/fpsyt.2021.657105.
[112] MA Y, ZHOU H, LI C P, et al. Differential metabolites in Chinese autistic children: a multi-center study based on urinary 1H-NMR metabolomics analysis[J]. Frontiers in Psychiatry, 2021, 12: 624767.DOI:10.3389/fpsyt.2021.624767.
[113] QU Y C, SU C J, ZHAO Q H, et al. Gut microbiota-mediated elevated production of secondary bile acids in chronic unpredictable mild stress[J]. Frontiers in Pharmacology, 2022, 13: 837543. DOI:10.3389/fphar.2022.837543.
[114] LIANG X Q, MAI P Y, QIN H, et al. Integrated 16S rRNA sequencing and metabolomics analysis to investigate the antidepressant role of Yang-Xin-Jie-Yu decoction on microbe-gut-metabolite in chronic unpredictable mild stress-induced depression rat model[J]. Frontiers in Pharmacology, 2022, 13: 972351. DOI:10.3389/fphar.2022.972351.
[115] CHEN T L, ZHOU K J, SUN T, et al. Altered bile acid glycine:taurine ratio in the progression of chronic liver disease[J]. Journal of Gastroenterology and Hepatology, 2022, 37(1): 208-215.DOI:10.1111/jgh.15709.
[116] HARNISCH L O, MIHAYLOV D, BEIN T, et al. A reduced glycineto-taurine ratio of conjugated serum bile acids signifies an adaptive mechanism and is an early marker of outcome in acute respiratory distress syndrome[J]. Internal and Emergency Medicine, 2023, 18(2):607-615. DOI:10.1007/s11739-022-03152-0.
[117] YU H N, GUO Z Z, SHEN S R, et al. Effects of taurine on gut microbiota and metabolism in mice[J]. Amino Acids, 2016, 48(7):1601-1617. DOI:10.1007/s00726-016-2219-y.
[118] QIAN W K, LI M Y, YU L L, et al. Effects of taurine on gut microbiota homeostasis: an evaluation based on two models of gut dysbiosis[J]. Biomedicines, 2023, 11(4): 1048. DOI:10.3390/biomedicines11041048.
[119] FANG H, MENG F P, PIAO F Y, et al. Effect of taurine on intestinal microbiota and immune cells in peyer’s patches of immunosuppressive mice[J]. Advances in Experimental Medicine and Biology, 2019, 1155:13-24. DOI:10.1007/978-981-13-8023-5_2.
[120] 欧阳轶强, 宋玉美, 梁柏莹, 等. 牛磺酸对乳腺癌大鼠肠道菌群的影响[J]. 广西医科大学学报, 2017, 34(4): 491-495. DOI:10.16190/j.cnki.45-1211/r.2017.04.003.
[121] 朱伏丽. 牛磺酸对非酒精性脂肪性肝病大鼠肝脏及肠黏膜屏障的防护作用[D]. 南宁: 广西医科大学, 2020: 47-48. DOI:10.27038/d.cnki.ggxyu.2020.000634.
[122] 钱嘉豪. 高糖饲料添加牛磺酸对不同食性鱼类IGF-1R SUMOylation修饰和糖代谢的影响研究[D]. 湛江: 广东海洋大学,2021: 29-44. DOI:10.27788/d.cnki.ggdhy.2021.000202.
[123] PENG M, LUO H J, KUMAR V, et al. Dysbiosis of intestinal microbiota induced by dietary oxidized fish oil and recovery of diet-induced dysbiosis via taurine supplementation in rice field eel(Monopterus albus)[J]. Aquaculture, 2019, 512: 734288. DOI:10.1016/j.aquaculture.2019.734288.
[124] HUANG Y H, CHANG L C, CHANG Y C, et al. Compositional alteration of gut microbiota in psoriasis treated with IL-23 and IL-17 inhibitors[J]. International Journal of Molecular Sciences, 2023, 24(5):4568. DOI:10.3390/ijms24054568.
[125] STACY A, ANDRADE-OLIVEIRA V, MCCULLOCH J A, et al.Infection trains the host for microbiota-enhanced resistance to pathogens[J]. Cell, 2021, 184(3): 615-627.e17. DOI:10.1016/j.cell.2020.12.011.
[126] GKOLFAKIS P, DIMITRIADIS G, TRIANTAFYLLOU K. Gut microbiota and non-alcoholic fatty liver disease[J]. Hepatobiliary &Pancreatic Diseases International, 2015, 14(6): 572-581. DOI:10.1016/s1499-3872(15)60026-1.
[127] ZHU W H, CHEN S W, CHEN R G, et al. Taurine and tea polyphenols combination ameliorate nonalcoholic steatohepatitis in rats[J].BMC Complementary and Alternative Medicine, 2017, 17(1): 455.DOI:10.1186/s12906-017-1961-3.
[128] KAESLER S, SOBIESIAK M, KNEILLING M, et al. Effective T-cell recall responses require the taurine transporter Taut[J]. European Journal of Immunology, 2012, 42(4): 831-841. DOI:10.1002/eji.201141690.
[129] SHIMIZU M, ZHAO Z H, ISHIMOTO Y, et al. Dietary taurine attenuates dextran sulfate sodium (DSS)-induced experimental colitis in mice[J]. Advances in Experimental Medicine and Biology, 2009,643: 265-271. DOI:10.1007/978-0-387-75681-3_27.
[130] LEVY M, THAISS C A, ZEEVI D, et al. Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling[J]. Cell, 2015, 163(6): 1428-1443.DOI:10.1016/j.cell.2015.10.048.