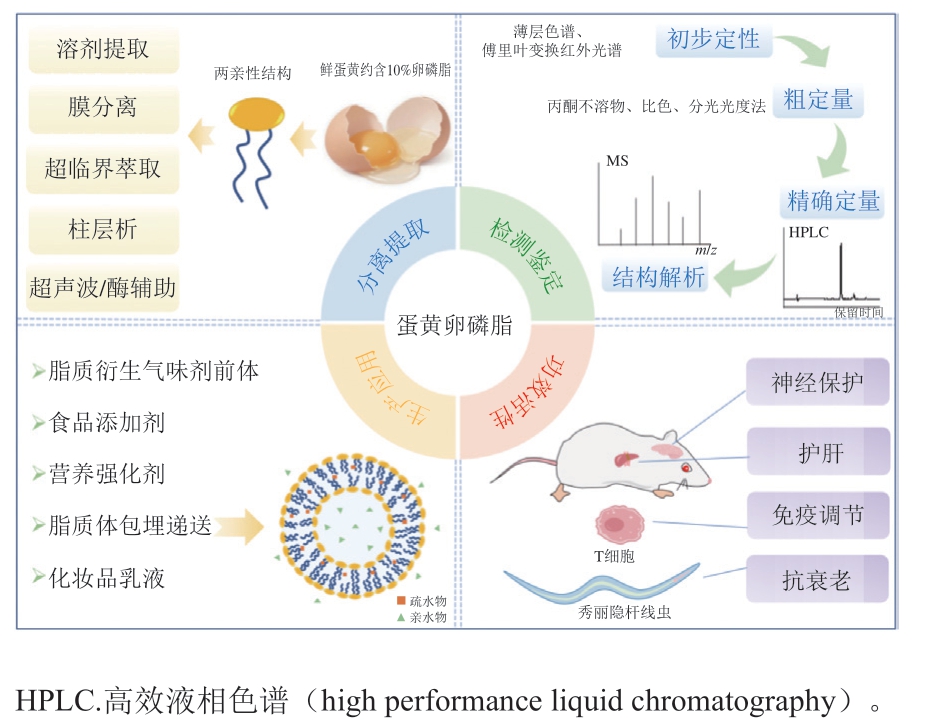
图1 蛋黄卵磷脂的提取鉴定、功效活性及应用
Fig. 1 Extraction, identification, bioactivities and applications of egg yolk lecithin
Advances in Research on Egg Yolk Lecithin: Extraction, Identification, Bioactivities and Applications
刘嘉贤, 阚启鑫, 胡伟, 等. 蛋黄卵磷脂的研究进展: 提取鉴定、功效活性及应用[J]. 食品科学, 2026, 47(2): 386-397.DOI:10.7506/spkx1002-6630-20250722-179. http://www.spkx.net.cn
LIU Jiaxian, KAN Qixin, HU Wei, et al. Advances in research on egg yolk lecithin: extraction, identification, bioactivities and applications[J]. Food Science, 2026, 47(2): 386-397. (in Chinese with English abstract) DOI:10.7506/spkx1002-6630-20250722-179. http://www.spkx.net.cn
我国是全球鸡蛋产业的核心生产国和消费国,2023年我国鸡蛋产量约为2 839万 t,同比增长1.3%[1]。虽然我国鸡蛋产量庞大,但当前鸡蛋还是以鲜食消费为主,深加工程度仅为7%左右,且加工蛋制品中约有80%为咸蛋、卤蛋、皮蛋、糟蛋等传统蛋制品,深加工程度远低于日本(55%)和美国(33%)[2]。鸡蛋主要由蛋壳、蛋清和蛋黄3 个部分构成,蛋黄占鲜蛋总质量近1/3,富含蛋黄油、卵黄高磷蛋白、卵黄免疫球蛋白、卵磷脂、胆固醇(cholesterol,TC)等多种生物活性成分[3-4],其应用价值远不止于传统的食用范畴。为了深入挖掘蛋黄的应用潜力,其全组分高值化利用已成为研究的热点。通过精准的分离提取技术,蛋黄中各活性组分能被单独分离,成为高附加值产品,从而实现资源的最大化利用和产业升级。例如,以鲜蛋黄为原料,蛋黄加水稀释离心后再盐析提纯能得到卵黄免疫球蛋白;剩余蛋黄颗粒经脱脂、盐溶,得到卵黄高磷蛋白;脱去的脂质组分用溶剂分级处理,得到蛋黄油和卵磷脂[4]。
鲜蛋黄中含约10%的卵磷脂,是提取动物源磷脂的理想原料[5]。当前大量研究证实,蛋黄卵磷脂具有保护神经[6]、护肝[7]、抗衰老[8-10]、免疫调节[11]、护肤[12]、药物递送[13]多重功效,可用于食品、药品、保健品、化妆品等领域,深加工潜力巨大。因此,亟待开发新型的卵磷脂提取加工工艺,深入挖掘蛋黄卵磷脂的功能活性,以提高禽蛋应用价值。基于此,本文对蛋黄卵磷脂的结构特性、提取鉴定、功能活性及应用进行全面叙述(图1),旨在能对蛋黄卵磷脂未来的研究和开发应用提供一定的理论参考。

图1 蛋黄卵磷脂的提取鉴定、功效活性及应用
Fig. 1 Extraction, identification, bioactivities and applications of egg yolk lecithin
蛋黄卵磷脂以主链结构划分,可分为以甘油或神经鞘氨醇为骨架两大类。甘油磷脂以甘油为主碳链,sn-1和sn-2位分别连接脂肪酸,sn-3位连接磷酸基团后再与不同极性头基酯化,形成磷脂酰胆碱(phosphatidylcholine,PC)、磷脂酰乙醇胺(phosphatidylethanolamine,PE)、磷脂酸(phospholipid acid,PA)、磷脂酰肌醇(phosphatidylinositol,PI)、磷脂酰丝氨酸(phosphatidylserine,PS)、磷脂酰甘油(phosphatidylglycerol,PG)等[14-16]。通常饱和脂肪酸多连接在sn-1位,不饱和脂肪酸连接在sn-2位[14]。甘油磷脂sn-1或sn-2位脂肪酸能被磷脂酶酶解生成羟基,形成溶血磷脂酰胆碱(lysophosphatidylcholine,LPC)、溶血磷脂酰乙醇胺(lysophosphatidylethanolamine,LPE)、溶血磷脂酸(lysophosphatidic acid,LPA)等[16]。鞘脂则以鞘氨醇为基本骨架,鞘氨醇的氨基与脂肪酸通过酰胺键连接生成神经酰胺(ceramide,Cer),再与不同极性头基酯化形成鞘脂,可进一步细分为鞘磷脂(sphingomyelin,SM)、酸性糖鞘脂、碱性糖鞘脂等[17]。蛋黄中的鞘脂以连接磷酸胆碱的SM为主,含量占总卵磷脂的1.0%~6.0%[18]。与其他来源磷脂相比,蛋黄卵磷脂的组成独特,PE、PC是其主要的两大组分,其中PC占60.0%~82.0%,PE占8.0%~26.0%,PS、PI、PA、SM和溶血磷脂等组分占比较少[5,15,18]。
蛋黄卵磷脂所连接的脂肪酸主要为棕榈酸C16:0(23.0%~31.9%)、亚油酸C18:2(11.4%~13.2%)、油酸C18:1(26.6%~29.4%)和硬脂酸C18:0(14.0%~18.3%),其中饱和脂肪酸占37.9%~51.0%,单不饱和脂肪酸占23.8%~32.7%,多不饱和脂肪酸占20.0%~30.1%[19-21]。蛋黄卵磷脂sn-1和sn-2位大多数通过酯键(—COO—)与脂肪酸相连,形成双酰基结构,如PC(16:1/18:0)[19]。但最近新的研究发现,部分蛋黄磷脂sn-1位可以通过醚键(—O—)与脂肪酸相连,生成烷基醚磷脂或缩醛磷脂[20,22-24]。烷基醚磷脂sn-1位以醚键与烷基链相连(如PC(O-16:1/18:0)),形成的饱和醚键结构能增强分子稳定性;而缩醛磷脂sn-1位醚键直接与双键相连,形成活泼的乙烯醚结构(如PE(P-16:1/18:1)),具有更好的抗氧化性[25-26]。
与普通的甘油三酯(triglyceride,TG)不同,卵磷脂含有亲水端的磷酸基团-极性头基和疏水端的脂肪酸链,是典型的两亲性物质,亲水亲脂平衡值为4.0~9.7[27]。卵磷脂在水中能形成胶束、脂质体、平面双层或六角相II结构,它的排列形式受极性头基大小、疏水链结构、温度、离子强度等因素影响,常用临界堆积参数(critical packing parameter,CPP)预测其自组装结构。溶血磷脂仅有一条脂肪酸链,极性头基覆盖表面积大,分子几何形状为倒锥形,CPP<1/3时形成球状胶束,1/3<CPP<1/2时自组装为棒状胶束。PC、PI、PS和SM的极性头基和疏水双链所占表面积相似,分子几何形状近似圆柱体,在1/2<CPP<1时倾向于形成闭合囊泡,CPP=1时形成平面双层结构。相比,PE的极性头基稍小,分子几何形状为锥形,CPP>1,形成六角相II结构[16,28]。蛋黄卵磷脂的具体结构性质见表1。
表1 蛋黄卵磷脂主要组分的结构性质
Table 1 Structural properties of the major components of egg yolk lecithin
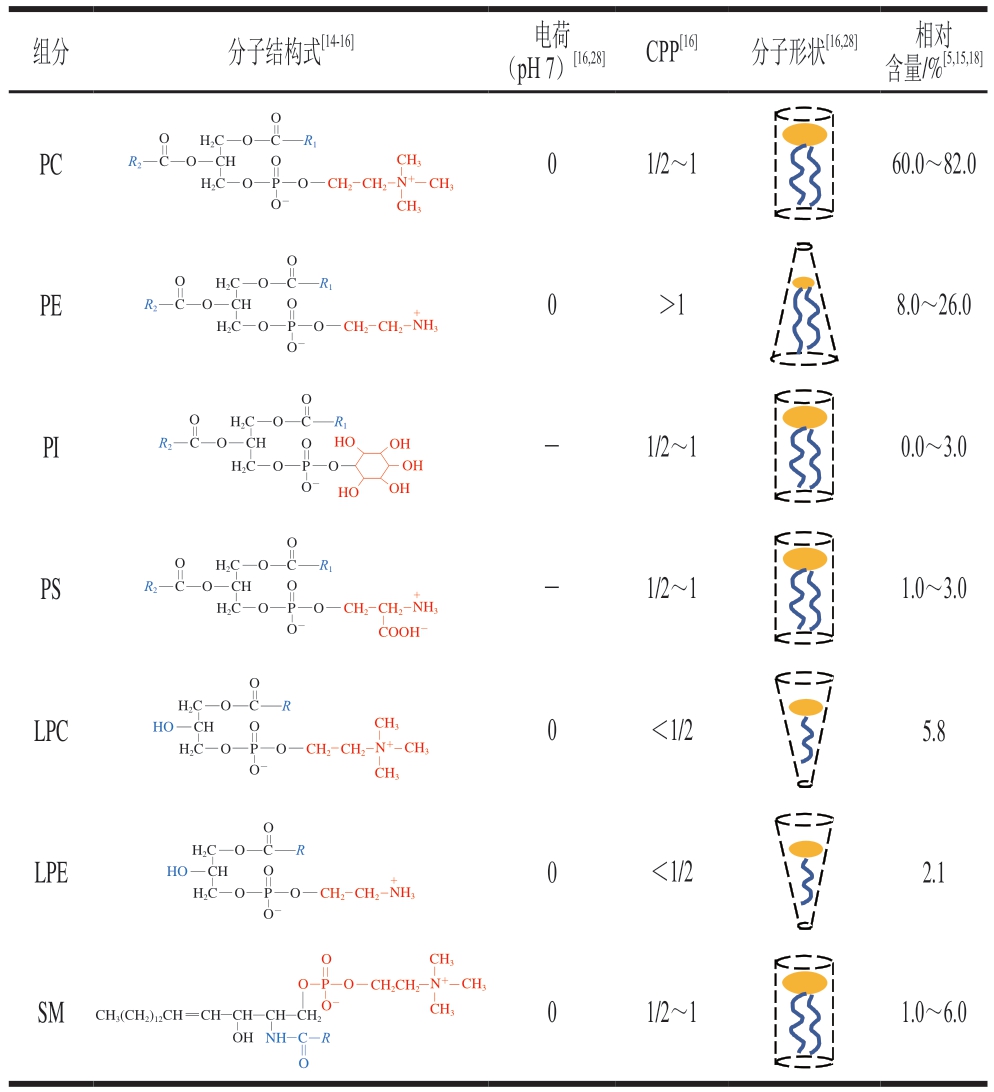
注:0.中性电荷;-.负电荷。
组分分子结构式[14-16]电荷(pH 7)[16,28]CPP[16]分子形状[16,28]相对含量/%[5,15,18]OOCH2CH2CH3 CH3 O C H2C R1 PC R2 O C O CH H2C O CH3 N+O P 01/2~160.0~82.0 O-O H2C C R1 PE R2 O C O CH H2C O +0>18.0~26.0 OOCH2CH2NH3 OPOO H2C C R1 PI R2 O C O CH H2C O HO OH -1/2~10.0~3.0 OO O P OH O-HO OH PS OC H2C O C R1 R2O CH H2C O +OOCH2CH2NH3 OP O-COOH--1/2~11.0~3.0 H2CR O C LPC HO CH H2C O CH3 OOCH2CH2N+CH3 OP O-CH3 H2CR O C+LPENH3 HO CH H2C O OOCH2CH2 OPO-0<1/25.8 0<1/22.1 O CH3 OP O SM CH3 CH2CH2N+CH3 CH3(CH2)12CHCHCHCHCH2 NHC O O-01/2~11.0~6.0 OH R
蛋黄卵磷脂提取常用到溶剂法、超临界萃取、膜分离、柱层析等手段,或加以超声波、酶辅助技术提高提取效率。根据卵磷脂的极性,蛋黄卵磷脂多用乙醇提取,再结合己烷脱油、丙酮沉淀、低温冬化等方法分离纯化[29-32]。尤其是利用卵磷脂不溶于丙酮的特性,经丙酮不同程度的洗涤,蛋黄卵磷脂纯度在40%~96%[29-32]。相比而言,超临界萃取的纯度更高,达90%以上,但得率较低,提取成本高[33]。膜分离法通过制备反向胶束截留分离,能有效纯化卵磷脂[34]。柱层析利用不同亚类卵磷脂在固定相(树脂、氧化铝、硅胶)的吸附能力不同进行洗脱,可以分离出高纯度的单个组分卵磷脂,但同时也存在载量少、耗时长的缺陷[35-37]。表2总结了现阶段蛋黄卵磷脂的分离提取工艺。
表2 蛋黄卵磷脂的分离提取工艺
Table 2 Separation and extraction processes of egg yolk lecithin
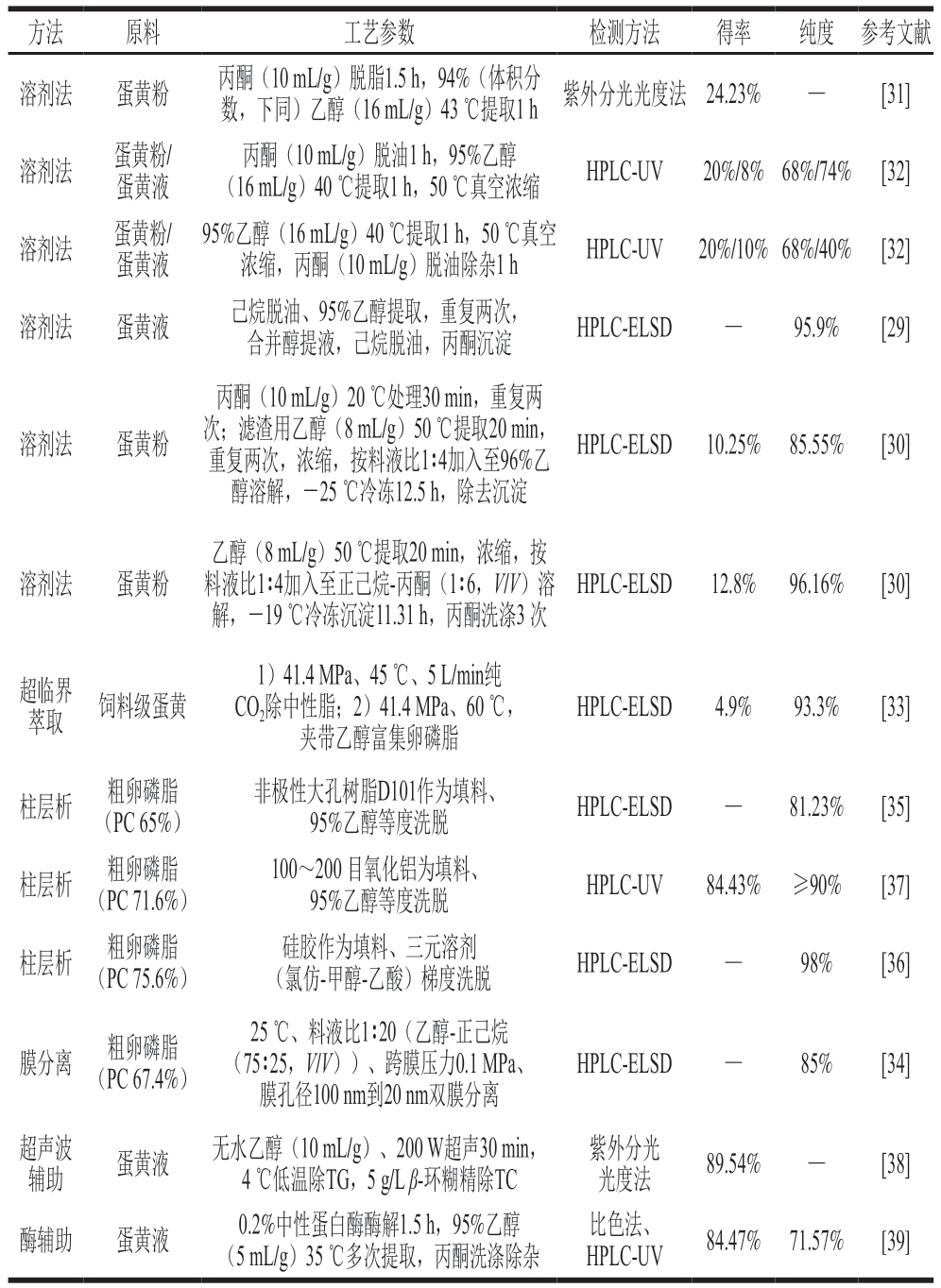
注:UV.紫外(ultraviolet);ELSD.蒸发光散射检测器(evaporative light-scattering detector);—.未查到。
方法原料工艺参数检测方法得率纯度参考文献溶剂法蛋黄粉丙酮(10 mL/g)脱脂1.5 h,94%(体积分数,下同)乙醇(16 mL/g)43 ℃提取1 h紫外分光光度法24.23%—[31]溶剂法蛋黄粉/蛋黄液丙酮(10 mL/g)脱油1 h,95%乙醇(16 mL/g)40 ℃提取1 h,50 ℃真空浓缩HPLC-UV20%/8%68%/74%[32]溶剂法蛋黄粉/蛋黄液95%乙醇(16 mL/g)40 ℃提取1 h,50 ℃真空浓缩,丙酮(10 mL/g)脱油除杂1 hHPLC-UV20%/10%68%/40%[32]溶剂法蛋黄液己烷脱油、95%乙醇提取,重复两次,合并醇提液,己烷脱油,丙酮沉淀HPLC-ELSD—95.9%[29]溶剂法蛋黄粉丙酮(10 mL/g)20 ℃处理30 min,重复两次;滤渣用乙醇(8 mL/g)50 ℃提取20 min,重复两次,浓缩,按料液比1∶4加入至96%乙醇溶解,-25 ℃冷冻12.5 h,除去沉淀HPLC-ELSD10.25%85.55%[30]溶剂法蛋黄粉乙醇(8 mL/g)50 ℃提取20 min,浓缩,按料液比1∶4加入至正己烷-丙酮(1∶6,V/V)溶解,-19 ℃冷冻沉淀11.31 h,丙酮洗涤3 次HPLC-ELSD12.8%96.16%[30]超临界萃取饲料级蛋黄1)41.4 MPa、45 ℃、5 L/min纯CO2除中性脂;2)41.4 MPa、60 ℃,夹带乙醇富集卵磷脂HPLC-ELSD4.9%93.3%[33]柱层析粗卵磷脂(PC 65%)非极性大孔树脂D101作为填料、95%乙醇等度洗脱HPLC-ELSD—81.23%[35]柱层析粗卵磷脂(PC 71.6%)100~200 目氧化铝为填料、95%乙醇等度洗脱HPLC-UV84.43%≥90%[37]柱层析粗卵磷脂(PC 75.6%)硅胶作为填料、三元溶剂(氯仿-甲醇-乙酸)梯度洗脱HPLC-ELSD—98%[36]膜分离粗卵磷脂(PC 67.4%)25 ℃、料液比1∶20(乙醇-正己烷(75∶25,V/V))、跨膜压力0.1 MPa、膜孔径100 nm到20 nm双膜分离HPLC-ELSD—85%[34]超声波辅助蛋黄液无水乙醇(10 mL/g)、200 W超声30 min,4 ℃低温除TG,5 g/L β-环糊精除TC紫外分光光度法89.54%—[38]酶辅助蛋黄液0.2%中性蛋白酶酶解1.5 h,95%乙醇(5 mL/g)35 ℃多次提取,丙酮洗涤除杂比色法、HPLC-UV84.47%71.57%[39]
溶剂法是利用目标物极性选择适宜溶剂的一种提取方法,具有成本低、易规模化操作的优势,是工业生产最常用的一种方法。卵磷脂提取常使用乙醇作为溶剂[40-41],结合己烷脱油、丙酮沉淀、低温冬化除中性脂等方法分离纯化[29-32]。虽然样品先用丙酮处理能有效除去中性脂和其他杂质的干扰,增加卵磷脂提取量[32,42],但丙酮的长期使用会对环境和人体造成威胁,从安全环保方面考虑,可选择用硫酸镁代替丙酮沉淀卵磷脂。与传统金属盐沉淀相比,硫酸镁在沉淀过程中不会形成磷脂酰胆碱盐,只与其他杂质结合,减少了后续从金属盐回收PC的程序[43]。
超临界CO2萃取通过在超临界状态下调整温度和压力,达到依次萃取不同组分的目的。在整个提取工艺中,样品在接近室温和充满CO2的环境下进行,适合易氧化、不耐高温的卵磷脂提取。由于CO2具有非极性和分子质量低的两大特点,只能萃取非极性或弱极性物质,对于极性较大的卵磷脂需要另外夹带极性溶剂提取[44-45]。乙醇因其绿色安全性(一般公认安全(Generally Recognized as Safe,GRAS)溶剂)一直是食品工业提取的理想溶剂,通过夹带乙醇能有效提高卵磷脂在超临界二氧化碳(supercritical CO2,SC-CO2)体系中的溶解性[46],并且在该工艺下提取出的卵磷脂各项检测指标(乳化性、稳定性、生物活性等)均比直接用乙醇浸泡提取工艺的更加优异[47-48]。虽然也有报道称用异丙醇作为助溶剂提取出的卵磷脂纯度更高[49],但考虑到安全环保性,目前大多数工艺还是选择用乙醇作为助溶剂。总体来说,利用SCCO2萃取蛋黄卵磷脂可分两大步进行:第一步先用纯CO2脱去蛋黄油脂;第二步再通过乙醇夹带提取[50]。若想要再进一步提高卵磷脂的纯度和回收率,可在此基础上采用超临界反溶剂技术深度纯化[51]。
膜分离法利用不同物质的粒径、分子质量差异使其选择性透过膜,是一种常见的物理分离手段。卵磷脂(600~800 Da)和TG(600~1 000 Da)的分子质量相近,理论上膜技术难以将两者有效分离。但卵磷脂是两亲性物质,具有亲水/疏水末端,能在非水介质中形成球状结构的反向胶束[52-53]。因此,卵磷脂的分离可以通过制备反向胶束,增大粒径,选择合适的膜孔径进行截留。近年来,聚醚砜膜、无机陶瓷膜、氧化石墨烯层状复合膜等不同类型的过滤膜都已成功用于截留不同来源卵磷脂[54-56],其中刘青松等[34]利用陶瓷双膜(膜孔径100 nm到20 nm)能有效将蛋黄PC纯度从67%提升至85%。虽然膜分离是一种有效的分离纯化手段,但在应用过程中膜污染依然是核心的技术难点。膜分离利用卵磷脂在非水介质形成反向胶束进行截留,容易在膜表面形成凝胶层以及堵塞膜内部多孔通道,导致渗透通量下降[56]。因此,生产中需要定期对膜进行清洁维护,以提供更好的分离性能。利用正己烷在低压(0.5 bar)、高速(5.0 ms―1)的条件下清洁膜,能在一定程度上恢复膜的渗透通量[57]。
柱层析利用目标物在两相间的分配系数不同,经多次分配洗脱,能依次将不同亚类卵磷脂分离,适合高纯度卵磷脂的制备。但是同时也存在分离耗时长、载量小、溶剂消耗大等缺陷。目前,利用柱层析分离蛋黄卵磷脂常用树脂、氧化铝或硅胶作为固定相填料,经程序洗脱后能够得到高纯度的单个组分卵磷脂[35-37]。
除了上述方法,还有一些辅助技术常被用于提高提取效率,如超声波辅助、酶辅助、集成提取技术等。超声波辅助利用超声波产生的空化、振动、热效应[58-59],能加速卵磷脂溶出,减少溶剂消耗,提高提取效率[60]。酶辅助利用酶将与卵磷脂结合的大分子蛋白质分解成小分子肽,促进卵磷脂析出,缩短提取时间[61]。严喜鸾等[39]通过在蛋黄液中添加0.2%中性蛋白酶(酶活力80 000 U/g)酶解1.5 h,再用乙醇提取,有效提升了蛋黄卵磷脂的纯度和得率。集成提取技术(如超声辅助酶提取技术、酶辅助超临界萃取技术、酶辅助微波萃取技术等)通过联合运用多个提取方法的优点,克服某单一提取的缺陷,能显著提高提取的得率和效率[62-63]。Barry等[64]综合运用了酶解、超滤和超临界萃取的优化集成工艺,有效将酪乳粉中卵磷脂的初始相对含量从1.3%富集至56.24%。
综上,不同分离提取工艺都有其明确的优缺点。在工业转化中,蛋黄卵磷脂的提取工艺直接决定了生产的运行成本和产品的市场定位。传统的溶剂提取前期设备投资少,主要的运行成本集中在溶剂消耗和溶剂回收上,随着环保政策收紧,企业负担的环保、安全压力日益增大,综合成本优势下降,主要面向中低端保健品、食品、饲料市场。超临界萃取前期设备投资高昂,运行成本主要基于设备折旧和能耗,但所得产品品质好、纯度高,主要瞄准高端保健品、医药、化妆品等高附加值领域。柱层析法虽然能分离出极高纯度的单个组分卵磷脂,但昂贵的层析介质耗损、载量低、生产周期长、溶剂消耗大等缺陷,决定了它只能作为终端的精制手段,主要面向尖端医药和科研领域。膜分离法加工过程中条件温和,工业化成本主要是膜组件的频繁清洗维护与更换,因受限于处理通量和稳定性,其尚未成为主流的分离提取技术。表3对上述不同分离提取方法的工业化成本进行了评估比较。
表3 蛋黄卵磷脂不同分离提取方法的工业化成本
Table 3 Industrialization costs of different separation and extraction methods for egg yolk lecithin
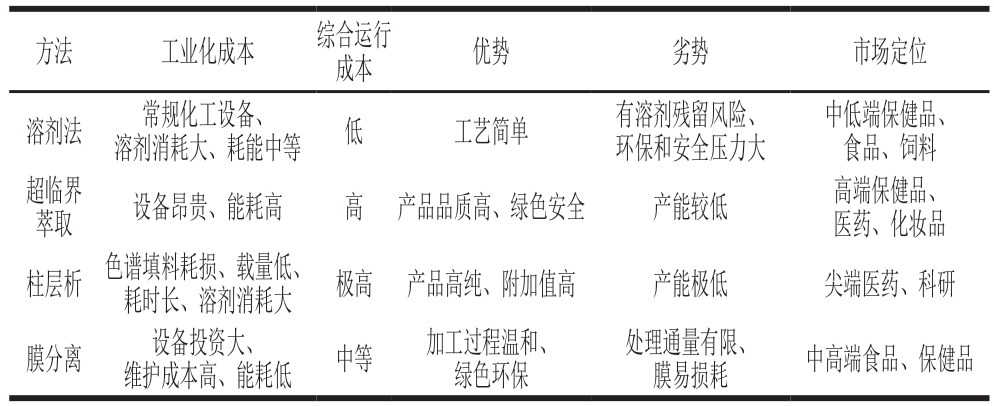
方法工业化成本综合运行成本优势劣势市场定位溶剂法常规化工设备、中低端保健品、食品、饲料超临界溶剂消耗大、耗能中等低工艺简单有溶剂残留风险、环保和安全压力大萃取设备昂贵、能耗高高产品品质高、绿色安全产能较低高端保健品、医药、化妆品柱层析色谱填料耗损、载量低、耗时长、溶剂消耗大极高产品高纯、附加值高产能极低尖端医药、科研膜分离设备投资大、维护成本高、能耗低中等加工过程温和、绿色环保处理通量有限、膜易损耗中高端食品、保健品
蛋黄卵磷脂的检测方法多样,适用于不同的检测需求。若要快速鉴定提取物是否为卵磷脂,可采用薄层色谱法(thin-layer chromatography,TLC)[65-66]或傅里叶变换红外光谱(Fourier transform infrared spectroscopy,FTIR)[38]进行初步的定性分析。丙酮不溶物法[67]、比色法[21]、分光光度法[38]适用于总卵磷脂的粗定量,测定结果往往包括其他干扰杂质,测定误差大。严格上说,粗定量方法不能直接用于表征样品中卵磷脂含量,还应进一步测定其纯度。
HPLC能对卵磷脂各组分精确定量,常配备UV检测器、ELSD、电雾式检测器(charged aerosol detector,CAD)或示差折光检测器(refractive index detector,RID)。RID利用流动相和样品之间折射率差异进行分析,对温度压力敏感,适合等度洗脱。由于卵磷脂各组分之间的极性具有一定差异,RID往往难以在合适的时间内将不同组分洗脱,分离度较差,因此该方法逐渐被淘汰[68]。目前,在卵磷脂测定中应用较为广泛的是UV检测器和ELSD。UV检测器具有响应时间短、线性范围广的优点,但卵磷脂处于紫外吸收末端(205~210 nm),容易受溶剂和环境干扰,导致检测灵敏度下降[69]。另外,基于特征发色基团的紫外吸收原理,卵磷脂sn-1、sn-2位连接不同脂肪酸时,所含官能团的数量、位置不同,容易引起同一组分不同脂肪酸组成的卵磷脂出峰时间存在差异,导致峰拖尾[35]。相比之下,ELSD是通用型检测器,它根据样品雾化蒸发后剩余颗粒物的光散射强度进行测定。ELSD在样品检测前就将流动相蒸发完全,不易受溶剂干扰,基线稳定,适合梯度洗脱,对卵磷脂各组分的分离度好[69]。由于ELSD对样品的响应信号是非线性的,需要利用对数转换或多项式拟合进行定量分析,样品定量相对繁琐。有研究对比了用UV和ELSD检测卵磷脂的效果,结果发现ELSD对卵磷脂的分离效果更好,峰形拖尾少,检测结果也更接近真实值[35,69]。CAD是高灵敏通用型检测器,检测限达纳克级,灵敏度远高于ELSD,对卵磷脂各组分的分离度好,弥补了通用型检测器灵敏度低的缺陷,是对ELSD的有益补充。有研究利用HPLC-CAD有效将PI、PE、PC、LPC、LPE、SM等组分分离,回收率为94.63%~101.53%,相对标准偏差为0.8%~1.98%,方法准确度高、重现性好[22]。除了检测器的区别,不同柱子和流动相对卵磷脂的分离也起决定作用。一般情况下,正相硅胶柱多用于分离不同亚类卵磷脂,C8、C18反相柱则适用于分离不同脂肪酸组成的卵磷脂。流动相常用到正己烷-异丙醇-水或乙腈-甲醇-水,通过加入适量的酸、碱或挥发性缓冲盐能有效改善色谱峰峰型,减少拖尾[70]。气相色谱(gas chromatography,GC)则用于检测卵磷脂的脂肪酸组成。卵磷脂经甲酯化处理后生成脂肪酸甲酯,然后通过极性毛细管柱分离,能有效检测鉴定出卵磷脂的脂肪酸构成[19-20]。
质谱分析和脂质组学通过高灵敏、高分辨率的质谱技术结合多维度的统计学分析和数据库在线比对(如Lipid Xplorer、Lipid Search、MS-DIAL、Lipid Maps)[71-72],能精确鉴定出复杂样品中的卵磷脂。在质谱分析方面,电喷雾电离(electrospray ionization,ESI)技术能有效保留卵磷脂的完整分子信息,通过串联质谱产生的特征碎片精确区分不同亚类磷脂[19-20,23-24]。PC、SM在[M+H]+正离子模式下产生m/z 184.07的磷酸胆碱头部碎片。PE、PS、PA、PI在[M-H]-负离子模式下更灵敏,PE通过分裂产生m/z 196.03的特征头部碎片确定,PS通过m/z 152.99碎片和87.03 Da中性丢失鉴定,PA因无特异性头部需依赖m/z 152.99的磷酸甘油骨架确定,PI通过m/z 241.01特征碎片确定[19,23,73]。上述不同亚类卵磷脂的特异性头基断裂方式为其结构解析提供了关键依据,后续再通过结合高分辨质谱提供的精确质量数可进一步推导卵磷脂的分子组成。脂质组学通过系统全面地分析这些数据,不仅能揭示卵磷脂脂肪酸的链长、不饱和度、链位异构等结构特征,还能解析其在生物体中的空间分布、代谢差异及潜在的生物学功能[74]。
虽然质谱本身是一个优秀的定性工具,但其在离子化过程中易受样品基质干扰,仪器信号响应强度难以真实反映样品浓度。基于此,并行检测技术很好地解决了磷脂同时定性定量分析的问题,如HPLC-CAD-MS/MS联用技术结合了色谱的分离能力、CAD通用定量能力以及MS/MS定性能力,在一次样品运行分析中能同步获得不同检测器的互补信息,实现对样品全面高效的表征[75]。表4总结了当前蛋黄卵磷脂不同检测方法的原理及优缺点。
表4 蛋黄卵磷脂不同检测方法的原理及优缺点
Table 4 Principles, advantages and disadvantages of different detection methods for egg yolk lecithin
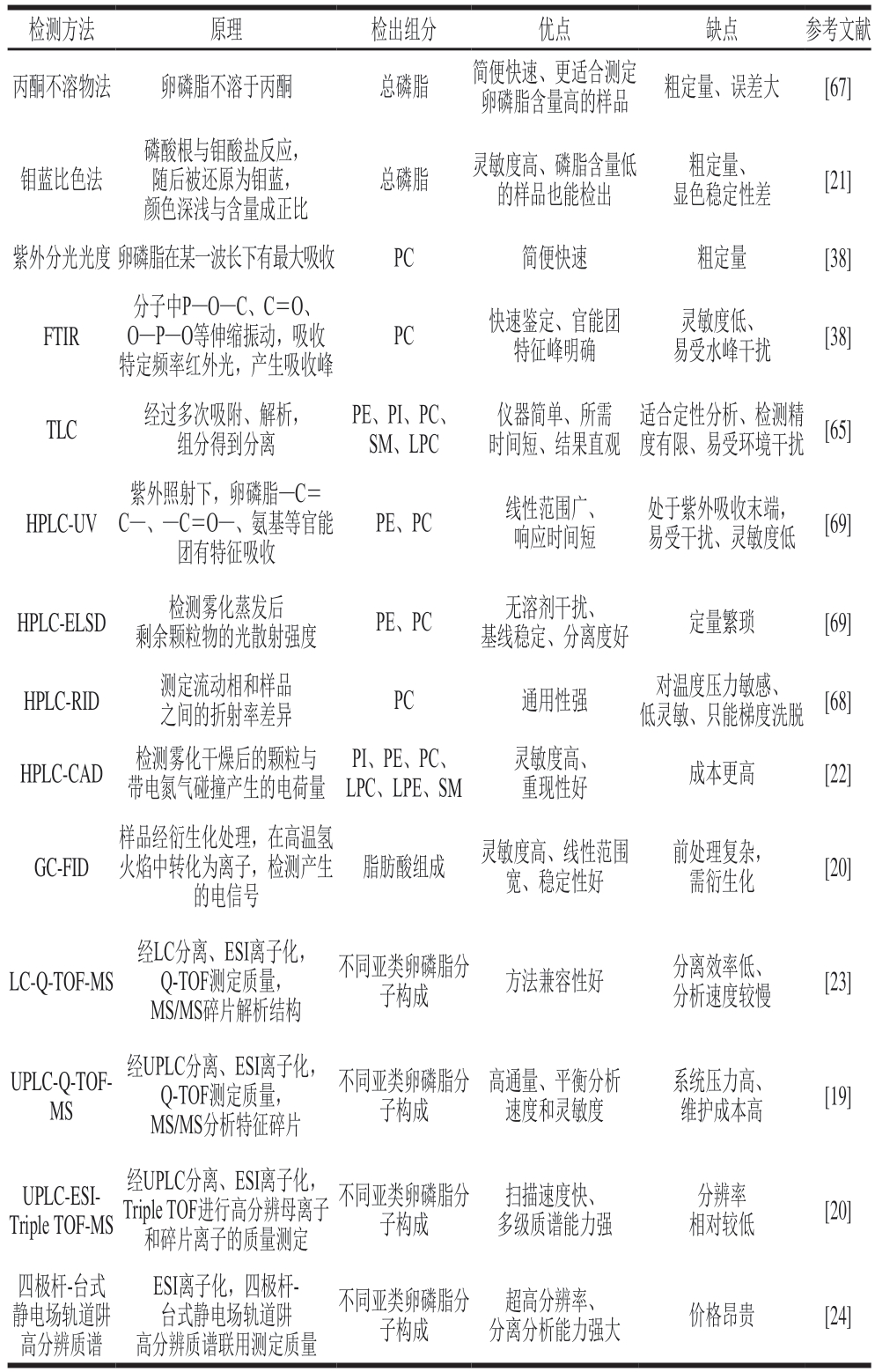
注:FID.火焰离子化检测器(flame ionization detector);LC-Q-TOFMS.液相色谱-四极杆-飞行时间质谱(liquid chromatography-quadrupoletime of flight-mass spectrometry);UPLC.超高效液相色谱(ultra-high performance liquid chromatography)。
检测方法原理检出组分优点缺点参考文献丙酮不溶物法卵磷脂不溶于丙酮总磷脂简便快速、更适合测定卵磷脂含量高的样品粗定量、误差大[67]钼蓝比色法磷酸根与钼酸盐反应,随后被还原为钼蓝,颜色深浅与含量成正比总磷脂灵敏度高、磷脂含量低的样品也能检出粗定量、显色稳定性差[21]紫外分光光度卵磷脂在某一波长下有最大吸收PC简便快速粗定量[38]FTIR分子中P—O—C、C=O、O—P—O等伸缩振动,吸收特定频率红外光,产生吸收峰PC快速鉴定、官能团特征峰明确灵敏度低、易受水峰干扰[38]TLC经过多次吸附、解析,组分得到分离PE、PI、PC、SM、LPC仪器简单、所需时间短、结果直观适合定性分析、检测精度有限、易受环境干扰[65]HPLC-UV紫外照射下,卵磷脂—C=C—、—C=O—、氨基等官能团有特征吸收PE、PC线性范围广、响应时间短处于紫外吸收末端,易受干扰、灵敏度低[69]HPLC-ELSD检测雾化蒸发后剩余颗粒物的光散射强度PE、PC无溶剂干扰、基线稳定、分离度好定量繁琐[69]HPLC-RID测定流动相和样品之间的折射率差异PC通用性强对温度压力敏感、低灵敏、只能梯度洗脱[68]HPLC-CAD检测雾化干燥后的颗粒与带电氮气碰撞产生的电荷量PI、PE、PC、LPC、LPE、SM灵敏度高、重现性好成本更高[22]GC-FID样品经衍生化处理,在高温氢火焰中转化为离子,检测产生的电信号脂肪酸组成灵敏度高、线性范围宽、稳定性好前处理复杂,需衍生化[20]LC-Q-TOF-MS经LC分离、ESI离子化,Q-TOF测定质量,MS/MS碎片解析结构不同亚类卵磷脂分子构成方法兼容性好分离效率低、分析速度较慢[23]UPLC-Q-TOFMS经UPLC分离、ESI离子化,Q-TOF测定质量,MS/MS分析特征碎片不同亚类卵磷脂分子构成高通量、平衡分析速度和灵敏度系统压力高、维护成本高[19]UPLC-ESITriple TOF-MS经UPLC分离、ESI离子化,Triple TOF进行高分辨母离子和碎片离子的质量测定不同亚类卵磷脂分子构成扫描速度快、多级质谱能力强分辨率相对较低[20]四极杆-台式静电场轨道阱高分辨质谱ESI离子化,四极杆-台式静电场轨道阱高分辨质谱联用测定质量不同亚类卵磷脂分子构成超高分辨率、分离分析能力强大价格昂贵[24]
蛋黄卵磷脂是一种天然的动物源磷脂复合物,由PC、PE、SM、PS等活性物质组成。其不仅是构成细胞膜双层结构的重要组分,还能通过促进乙酰胆碱(acetylcholine,Ach)合成、抑制氧化应激、参与脂质代谢调控、调节信号传导等途径,发挥保护神经[76-77]、护肝[7,78]、抗衰老[8-10]、免疫调节[11]多重功效。
随着年龄增长,机体自身胆碱含量逐渐下降,容易引起脑神经退化,引发阿尔茨海默病(Alzheimer’s disease,AD)。AD是常见的神经性认知障碍疾病,其发病机理与β-淀粉样蛋白(amyloid β-protein,Aβ)沉积、胆碱能系统缺陷、氧化应激等密切相关。这些通路之间相互交织,共同诱导AD的发生。Aβ沉积不仅对神经元产生直接毒性作用,还会干扰胆碱能信号通路,加剧胆碱能神经元结构和功能的损伤。PC是构成神经元膜的重要成分,也是生成Ach的重要前体[79]。PC能有效维持神经元膜完整性和突触可塑性,抑制Aβ聚集,激活胆碱能神经元,促进Ach合成,缓解脑老化等相关认知障碍[6,80-81]。多项研究证实,蛋黄磷脂能修复由东莨菪碱诱导引起的胆碱能损伤和氧化应激,发挥神经保护作用[36,77]。近年来,新的研究发现不同构型蛋黄PC对治疗AD小鼠神经健康的效果有所差异,高不饱和PC(不饱和度53.3%)对提高小鼠学习和记忆的干预能力明显优于低不饱和PC(不饱和度41.2%)[76]。这一发现提示了蛋黄磷脂酰基链的不同饱和度可能会对其生物活性产生显著影响,未来关于蛋黄卵磷脂的构效研究值得进一步深入探究。
日常膳食中,长期摄入高糖高脂食物容易诱发机体出现非酒精性脂肪肝病(non-alcoholic fatty liver disease,NAFLD),其发生与肝脏脂肪过度堆积有关,可进一步恶化为非酒精性脂肪性肝炎(non-alcoholic steatohepatitis,NASH)和肝硬化,甚至引起肝细胞癌、肝衰竭[82-83]。补充SM能有效减少肝脏对脂肪酸的摄取和储存,改善肝脏脂质代谢[84]。通过连续10 周饲喂高脂饮食小鼠0.1%的蛋黄SM,能降低小鼠肝脏TG、TC水平,抑制过氧化物酶体增殖物激活受体γ(peroxisome proliferator-activated receptor gamma,PPARγ)相关基因表达(如下调硬脂酰辅酶A去饱和酶1、脂肪酸转运蛋白、脂肪酸结合蛋白4基因),在预防肝脂肪变性方面表现出比牛奶SM更好的效果[78]。NAFLD进一步恶化为NASH的重要诱因是机体出现肝脏炎症反应[83],PC通过调节肝受体同源物-1、PPARγ、核因子κB(nuclear factor kappa B,NF-κB)等信号表达,能有效减轻脂肪性肝炎的发展,发挥护肝作用[85-86]。Yin Mingyu等[7]对大豆磷脂和蛋黄磷脂改善大鼠肝损伤的效果进行研究,发现蛋黄磷脂在改善肝脂肪变性、抗炎、抵御氧化应激、改善肝脏血脂谱等方面发挥出更好的效果,这说明了蛋黄磷脂在肝脏保护和相关代谢调控方面可能更具优势。
机体衰老是一个涉及多个方面的复杂生理过程,与DNA损伤、膜衰老、干细胞衰竭、自由基堆积等方面有关[87]。卵磷脂是构成细胞膜的重要组分,通过补充卵磷脂能维持膜完整性,延缓细胞衰老[88]。蛋黄卵磷脂有良好的自由基清除能力,研究表明6 mg/mL PE对1,1-二苯基-2-三硝基苯肼(1,1-diphenyl-2-picrylhydrazyl,DPPH)的清除率达69.15%[89],能减少体内自由基的堆积。PC、PE、PS能有效延长秀丽隐杆线虫(Caenorhabditis elegans)寿命,提高其对氧化应激的抵抗力,三者作用机制相似,均与激素效应和调节胰岛素/胰岛素样生长因子-1(insulin like growth factor-1,IGF-1)信号通路有关。一方面,磷脂通过诱导低水平活性氧,激活了相关应激反应和基因的表达(如热休克蛋白16.2和超氧化物歧化酶3上调),增强了对氧化应激的抵抗力。另一方面,磷脂通过抑制调节细胞生长的胰岛素/IGF-1通路并依赖DAF-16因子显著延长了线虫寿命[8-10]。
磷脂代谢在调节免疫细胞功能中起重要作用,增加PC(16:0/18:2)水平有助于调控辅助性T细胞(T helper cell,Th)17/调节性T细胞平衡,减轻自身免疫疾病中的炎症反应,维持免疫稳态[90]。通过补充蛋源PC能有效调节高脂大鼠的T细胞功能障碍[91]、改善肠道通透性[92]。近年来,有研究进一步探究了不同来源PC在调节T细胞功能障碍上作用机制的差异,结果发现大豆PC对肠道相关免疫激活表现出更强的调节作用,更能促进肠系膜淋巴结和脾细胞Th1型细胞因子(白细胞介素-2(interleukin-2,IL-2)、TNF-α)的分泌;鸡蛋PC则侧重促进抗炎因子IL-10的产生,增强Th2型免疫应答,有效缓解高脂饮食诱导的慢性炎症[11]。这一发现说明了不同来源PC在机体免疫调节中可能具有独特的调控特性。表5汇总了蛋黄卵磷脂的部分功效活性及机理。
表5 蛋黄卵磷脂的功效活性及机理
Table 5 Bioactivities and action mechanisms of egg yolk lecithin
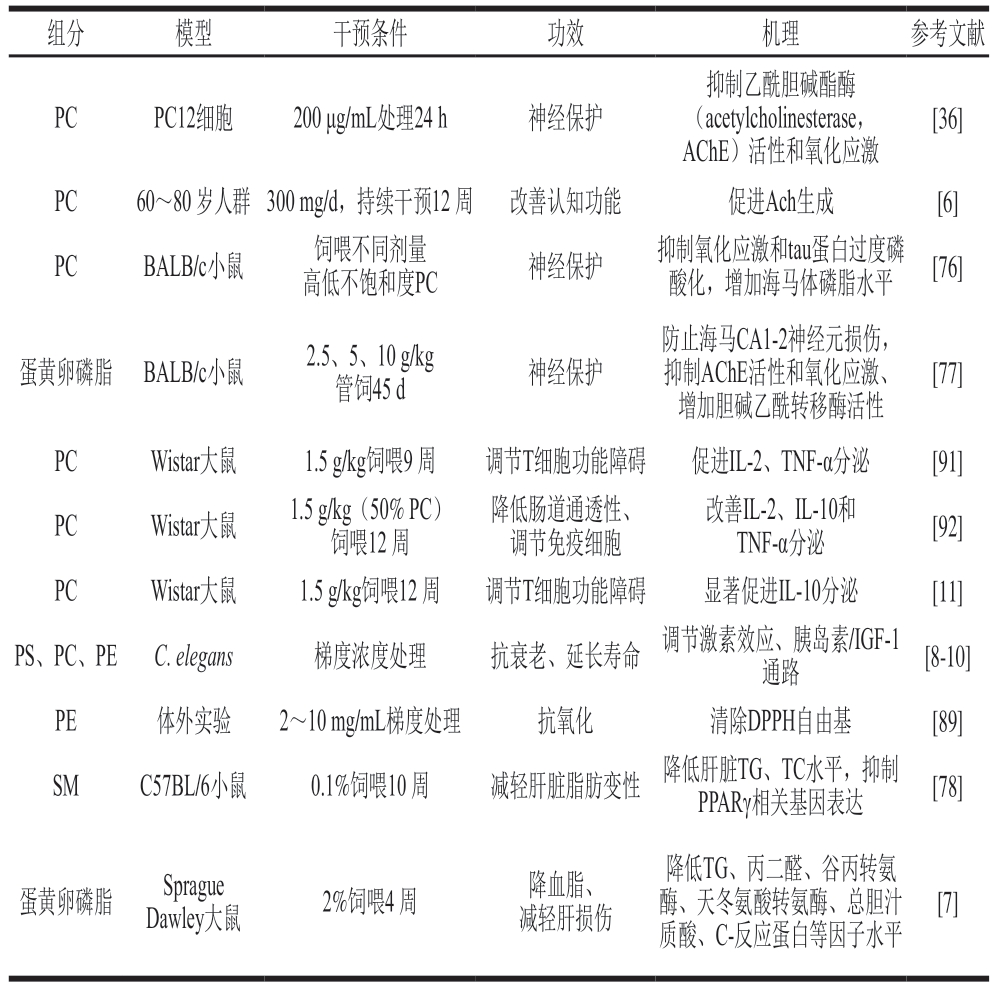
组分模型干预条件功效机理参考文献PCPC12细胞200 μg/mL处理24 h神经保护抑制乙酰胆碱酯酶(acetylcholinesterase,AChE)活性和氧化应激[36]PC60~80 岁人群300 mg/d,持续干预12 周改善认知功能促进Ach生成[6]PCBALB/c小鼠饲喂不同剂量高低不饱和度PC神经保护抑制氧化应激和tau蛋白过度磷酸化,增加海马体磷脂水平[76]蛋黄卵磷脂BALB/c小鼠2.5、5、10 g/kg管饲45 d神经保护防止海马CA1-2神经元损伤,抑制AChE活性和氧化应激、增加胆碱乙酰转移酶活性[77]PCWistar大鼠1.5 g/kg饲喂9 周调节T细胞功能障碍促进IL-2、TNF-α分泌[91]PCWistar大鼠1.5 g/kg(50% PC)饲喂12 周降低肠道通透性、调节免疫细胞改善IL-2、IL-10和TNF-α分泌[92]PCWistar大鼠1.5 g/kg饲喂12 周调节T细胞功能障碍显著促进IL-10分泌[11]PS、PC、PEC. elegans梯度浓度处理抗衰老、延长寿命调节激素效应、胰岛素/IGF-1通路[8-10]PE体外实验2~10 mg/mL梯度处理抗氧化清除DPPH自由基[89]SMC57BL/6小鼠0.1%饲喂10 周减轻肝脏脂肪变性降低肝脏TG、TC水平,抑制PPARγ相关基因表达[78]蛋黄卵磷脂Sprague Dawley大鼠2%饲喂4 周降血脂、减轻肝损伤降低TG、丙二醛、谷丙转氨酶、天冬氨酸转氨酶、总胆汁质酸、C-反应蛋白等因子水平[7]
蛋黄卵磷脂因其独特的分子结构和多样的生理功效,被广泛用于食品、保健品、医药、化妆品领域。在食品生产中,蛋黄卵磷脂可作为添加剂、营养强化剂或脂质衍生气味剂的前体使用。基于蛋黄卵磷脂的两亲性结构和优越的乳化性能,可将其制备成脂质体或化妆品乳液。利用脂质体的包埋作用,脂质体递送技术不仅能有效提高易敏活性物质或小分子药物的生物利用度及稳定性,还能实现目标物的精准靶向递送。
蛋黄卵磷脂独特的两亲性结构使其具有乳化、保湿、质构改善等功能,可用于加工生产巧克力[93]、鱼糜[94-96]、沙拉酱等制品,能有效改善产品的稳定性和品质。但在实际应用中,蛋源磷脂受成本、过敏风险、植物源磷脂竞争等方面的影响,其仅作为食品添加剂的应用较少,更多是作为营养强化剂使用。例如,在婴幼儿配方奶粉生产中,蛋黄卵磷脂具有植物源卵磷脂不可替代的优势。蛋黄卵磷脂具有更高含量的PC和SM,磷脂谱组成更接近母乳[97],在模拟母乳营养、促进婴幼儿大脑发育、维持配方乳稳定性[98-99]、改善消化吸收[100-101]等方面表现出显著优势。
近年来,新的研究发现蛋黄卵磷脂在加热过程中通过脂质氧化分解会生成大量挥发性风味物质,能显著增强食物中的油炸风味[102]和鸡肉香气[103]。蛋黄卵磷脂生成风味物质的关键是分子中所连接的亚油酸,亚油酸经氧化降解会生成9-羟基-10,12-十八碳二烯酸(9-hydroperoxy-10,12-octadecadienoic acid,9-HPOD),9-HPOD再进一步热加工转化为2,4-癸二烯醛,从而赋予食品强烈的脂肪香、油炸香和鸡肉香[104]。
卵磷脂与维生素、蛋白质并列为“第三营养素”,是维持人体健康不可缺少的关键成分,具有保护神经[6]、护肝[7]、抗衰老[8-10]、免疫调节[11]多重功效。大多数磷脂在肠道中被胰磷脂酶A2水解sn-2位酯键,生成溶血磷脂和游离脂肪酸,随后在肠上皮细胞重新酯化为磷脂(约20%磷脂可直接被动吸收),部分游离脂肪酸与TG再结合。新合成的磷脂和TG被整合成乳糜微粒,通过淋巴系统进入血液。当富含TG的颗粒被分解后,磷脂被高密度脂蛋白(high-density lipoprotein,HDL)吸收,然后经HDL介导,磷脂被输送至体内多个组织器官发挥作用[18,105]。
另外,基于两亲性的分子结构,卵磷脂能在水中自组装形成脂质体。卵磷脂的亲水基朝外、疏水链朝内,形成闭合的双分子层囊泡结构,亲水性物质被包裹在内部水室、疏水性物质镶入磷脂双分子层,常用于包埋易敏活性物质(如酚类、活性肽、植物提取物等),有利于提高活性物的稳定性和生物利用度[21,106-109]。
在负载药物方面,脂质体凭借良好的生物相容性和对小分子药物的包载能力,不仅能有效提高药物的生物利用度,还能实现精准的靶向递送[110-112]。负载阿霉素的天然卵黄脂质纳米载体能有效增强药物在肿瘤部位的富集,抑制肿瘤生长。经叶酸修饰后,卵黄脂质纳米载体还能进行主动的靶向递送,进一步促进药物富集[13]。目前,市面上已有多款基于脂质体包埋递送的药物,如两性霉素B、布比卡因、紫杉醇、柔红霉素/阿糖胞苷等[113]。两性霉素B是一种强效、广谱的抗真菌药物,同时也具有很强的肾毒性。利用脂质体包埋技术,两性霉素B经包埋后能有效减少药物在肾脏中的暴露,降低毒性[114]。另一种麻醉药物布比卡因通过采用多囊脂质体技术能实现缓慢、持久地释放,从而提供长效的局部镇痛疗效[115]。
卵磷脂具有保湿、维持肌肤嫩滑、修复皮肤屏障、促进活性物质渗透多重功效,被广泛应用在化妆品领域[116]。多项研究表明,利用脂质体包埋铜三肽[117]、水飞蓟素[118]、玫瑰果提取物[119]、维生素[120]等活性物,能有效保留物质的功效活性和稳定性,增强渗透。
蛋黄卵磷脂因其优越的乳化性能,可用于制备化妆品乳液。不同亚类卵磷脂的分子结构会影响乳液的自发曲率(H0),形成W/O或O/W乳液[121]。PC的极性头基截面积大于疏水尾部截面积,界面自然弯曲到油相(H0>0),油相以小液滴形式分散在水相中,形成O/W体系,适用于生产水基护肤品,具有肤感清爽、易清洗的优点。Kim等[12]利用脂质体包埋益生菌提取物,配制成O/W化妆水乳液,其表现出优异的抗氧化、防腐功效。相反,PE极性头基截面积小于疏水尾部截面积,易形成W/O体系,具有封闭性强、滋润度高等特点,适合生产长效保湿、防水防晒、卸妆去污[122]的化妆产品。蛋黄卵磷脂含有60.0%~82.0% PC,PC含量远大于PE[5,15,18],更适合制备O/W乳液[121]。
蛋黄卵磷脂是一种天然的功能性磷脂,具有独特的两亲性结构和多样的功效活性,其高值化利用主要集中在高纯度和高附加值产品上,聚焦于高端医药、保健品、化妆品领域。虽然近些年随着分离提取技术的不断完善,我国蛋黄卵磷脂的制备工艺取得了明显突破,但在工业化生产中其仍面临着高纯度提取技术壁垒高、产业链不完善、低端产能过剩、高端供给不足等瓶颈,高纯度卵磷脂依赖进口依然我国卵磷脂产业面临的核心痛点。虽然目前国内白云山汉方已突破了注射级卵磷脂产业化生产技术,但与国外相比仍存在一定差距,Lipoid GmbH、Fresenius Kabi、Avanti Polar Lipids等公司仍是垄断卵磷脂高端市场的核心企业。
未来蛋黄卵磷脂的研究应进一步聚焦于高效绿色的集成提取技术,突破高纯度纯化的技术瓶颈。同时推动蛋黄全组分高值化利用,充分利用剩余副产物,如提取完卵磷脂的蛋黄残渣可进一步用于提取卵黄高磷蛋白或酶解制备成蛋黄蛋白肽。在功效活性方面,虽然已有大量研究证明,蛋黄卵磷脂具有神经保护、护肝、抗衰老、免疫调节等多重功效,但其分子层面的调控机制和临床转化应用仍需继续深入研究验证,后续应进一步联合多组学分析揭示不同亚类卵磷脂的生物活性机制及构效关系,实现精准营养干预。蛋黄卵磷脂独特的两亲性结构,适合作为易敏活性物或小分子药物的高效载体,能够实现目标包埋物的精准靶向释放,在生物医药领域展现出独特的价值。随着多学科的交融发展,蛋黄卵磷脂正在不断突破传统的应用边界,展现出巨大的发展潜力。
[1] 孙从佼, 于爱芝, 汪洋, 等. 2023年蛋鸡产业发展情况、未来发展趋势及建议[J]. 中国畜牧杂志, 2024, 60(3): 307-311. DOI:10.19556/j.0258-7033.20240213-01.
[2] 范梅华, 王晓峰. 变革调整期, 我国蛋品加工行业高质量发展方向探讨[J]. 中国禽业导刊, 2021, 38(11): 2-13.
[3] HUANG X, AHN D U. How can the value and use of egg yolk be increased?[J]. Journal of Food Science, 2019, 84(2): 205-212.DOI:10.1111/1750-3841.14430.
[4] 郭晓徐. 蛋黄中天然活性成分连续提取技术研究[D]. 邯郸: 河北工程大学, 2022. DOI:10.27104/d.cnki.ghbjy.2022.000524.
[5] ZHAO F, LI R J, LIU Y, et al. Perspectives on lecithin from egg yolk: extraction, physicochemical properties, modification, and applications[J]. Frontiers in Nutrition, 2022, 9: 1082671. DOI:10.3389/fnut.2022.1082671.
[6] YAMASHITA S, KAWADA N, WANG W, et al. Effects of egg yolk choline intake on cognitive functions and plasma choline levels in healthy middle-aged and older Japanese: a randomized double-blinded placebo-controlled parallel-group study[J]. Lipids in Health and Disease, 2023, 22(1): 75. DOI:10.1186/s12944-023-01844-w.
[7] YIN M Y, MATSUOKA R, XI Y C, et al. Comparison of egg yolk and soybean phospholipids on hepatic fatty acid profile and liver protection in rats fed a high-fructose diet[J]. Foods, 2021, 10(7): 1569.DOI:10.3390/foods10071569.
[8] KIM B K, PARK S K. Phosphatidylserine modulates response to oxidative stress through hormesis and increases lifespan via DAF-16 in Caenorhabditis elegans[J]. Biogerontology, 2020, 21(2): 231-244.DOI:10.1007/s10522-020-09856-0.
[9] KIM S H, KIM B K, PARK S, et al. Phosphatidylcholine extends lifespan via DAF-16 and reduces amyloid-beta-induced toxicity in Caenorhabditis elegans[J]. Oxidative Medicine and Cellular Longevity, 2019, 2019(1): 2860642. DOI:10.1155/2019/2860642.
[10] PARK S, KIM B K, PARK S K. Supplementation with phosphatidylethanolamine confers anti-oxidant and anti-aging effects via hormesis and reduced insulin/IGF-1-like signaling in C. elegans[J].Mechanisms of Ageing and Development, 2021, 197: 111498.DOI:10.1016/j.mad.2021.111498.
[11] RUSNAK T, AZARCOYA-BARRERA J, MAKAROWSKI A, et al.Plant- and animal-derived dietary sources of phosphatidylcholine have differential effects on immune function in the context of a high-fat diet in male Wistar rats[J]. The Journal of Nutrition, 2024, 154(6): 1936-1944. DOI:10.1016/j.tjnut.2024.04.004.
[12] KIM H T, LEE J, JO Y J, et al. Application of liposome encapsulating Lactobacillus curvatus extract in cosmetic emulsion lotion[J].Materials, 2021, 14(24): 7571. DOI:10.3390/ma14247571.
[13] TANG Z, LUO C, JUN Y L, et al. Nanovector assembled from natural egg yolk lipids for tumor-targeted delivery of therapeutics[J]. ACS Applied Materials & Interfaces, 2020, 12(7): 7984-7994. DOI:10.1021/acsami.9b22293.
[14] SUN N, CHEN J, WANG D, et al. Advance in food-derived phospholipids: sources, molecular species and structure as well as their biological activities[J]. Trends in Food Science & Technology, 2018,80: 199-211. DOI:10.1016/j.tifs.2018.08.010.
[15] ALHAJJ M J, MONTERO N, YARCE C J, et al. Lecithins from vegetable, land, and marine animal sources and their potential applications for cosmetic, food, and pharmaceutical sectors[J].Cosmetics, 2020, 7(4): 87. DOI:10.3390/cosmetics7040087.
[16] WAGHULE T, SAHA R N, ALEXANDER A, et al. Tailoring the multi-functional properties of phospholipids for simple to complex self-assemblies[J]. Journal of Controlled Release, 2022, 349: 460-474.DOI:10.1016/j.jconrel.2022.07.014.
[17] YANG F, CHEN G X. The nutritional functions of dietary sphingomyelin and its applications in food[J]. Frontiers in Nutrition,2022, 9: 1002574. DOI:10.3389/fnut.2022.1002574.
[18] LORDAN R, TSOUPRAS A, ZABETAKIS I. Phospholipids of animal and marine origin: structure, function, and anti-inflammatory properties[J]. Molecules, 2017, 22(11): 1964. DOI:10.3390/molecules22111964.
[19] ALI A H, ZOU X Q, LU J, et al. Identification of phospholipids classes and molecular species in different types of egg yolk by using UPLCQ-TOF-MS[J]. Food Chemistry, 2017, 221: 58-66. DOI:10.1016/j.foodchem.2016.10.043.
[20] ZHOU L, YANG F, ZHAO M J, et al. Determination and comparison of phospholipid profiles in eggs from seven different species using UHPLC-ESI-Triple TOF-MS[J]. Food Chemistry, 2021, 339: 127856.DOI:10.1016/j.foodchem.2020.127856.
[21] BERNARDO J, VIDEIRA R A, VALENTÃO P, et al. Extraction of phospholipid-rich fractions from egg yolk and development of liposomes entrapping a dietary polyphenol with neuroactive potential[J]. Food and Chemical Toxicology, 2019, 133: 110749.DOI:10.1016/j.fct.2019.110749.
[22] WEI J L, ZHAO X, WANG S Q, et al. Determination of related substances in egg yolk lecithin by HPLC-CAD and characterization of its profiling by HPLC-Q-TOF-MS[J]. Journal of Pharmaceutical and Biomedical Analysis, 2022, 221: 115079. DOI:10.1016/j.jpba.2022.115079.
[23] XIE Y, WU B F, WU Z Y, et al. Ultrasound-assisted one-phase solvent extraction coupled with liquid chromatography-quadrupole time-offlight mass spectrometry for efficient profiling of egg yolk lipids[J]. Food Chemistry, 2020, 319: 126547. DOI:10.1016/j.foodchem.2020.126547.
[24] WOOD P L, MUIR W, CHRISTMANN U, et al. Lipidomics of the chicken egg yolk: high-resolution mass spectrometric characterization of nutritional lipid families[J]. Poultry Science, 2021, 100(2): 887-899.DOI:10.1016/j.psj.2020.11.020.
[25] ALMSHERQI Z A. Potential role of plasmalogens in the modulation of biomembrane morphology[J]. Frontiers in Cell and Developmental Biology, 2021, 9: 673917. DOI:10.3389/fcell.2021.673917.
[26] DEAN J M, LODHI I J. Structural and functional roles of ether lipids[J]. Protein & Cell, 2018, 9(2): 196-206. DOI:10.1007/s13238-017-0423-5.
[27] VAN HOOGEVEST P. Review: an update on the use of oral phospholipid excipients[J]. European Journal of Pharmaceutical Sciences, 2017, 108: 1-12. DOI:10.1016/j.ejps.2017.07.008.
[28] LI J, WANG X L, ZHANG T, et al. A review on phospholipids and their main applications in drug delivery systems[J]. Asian Journal of Pharmaceutical Sciences, 2015, 10(2): 81-98. DOI:10.1016/j.ajps.2014.09.004.
[29] PALACIOS L E, WANG T. Egg-yolk lipid fractionation and lecithin characterization[J]. Journal of the American Oil Chemists’ Society,2005, 82(8): 571-578. DOI:10.1007/s11746-005-1111-4.
[30] 黄春玲. 蛋黄磷脂提取及氧化铝吸附研究[D]. 无锡: 江南大学,2022. DOI:10.27169/d.cnki.gwqgu.2022.001232.
[31] 朱帅. 蛋黄磷脂酰胆碱的提取与酶法改性[D]. 北京: 北京化工大学,2022. DOI:10.26939/d.cnki.gbhgu.2022.000964.
[32] LIU Y, ZHU S, LIU X Z. Effect of four extraction processes with acetone-ethanol protocols on the properties of egg yolk phosphatidylcholine and the structure of residual protein[J]. Journal of Oleo Science, 2023, 72(1): 49-58. DOI:10.5650/jos.ess22219.
[33] SHAH A, AKOH C C, TOLEDO R T, et al. Isolation of a phospholipid fraction from inedible egg[J]. The Journal of Supercritical Fluids,2004, 30(3): 303-313. DOI:10.1016/j.supflu.2003.09.010.
[34] 刘青松, 王文高, 丁丹华, 等. 膜分离纯化蛋黄卵磷脂工艺研究[J].粮食与油脂, 2011, 24(6): 20-23.
[35] 田育苗. 蛋黄卵磷脂纯化工艺研究[D]. 北京: 北京化工大学, 2013:35-46.
[36] CHEN J, LIN S Y, SUN N, et al. Egg yolk phosphatidylcholine:extraction, purification and its potential neuroprotective effect on PC12 cells[J]. Journal of Functional Foods, 2019, 56: 372-383. DOI:10.1016/j.jff.2019.03.037.
[37] 龚金炎, 靳羽晓, 王静静, 等. 氧化铝柱层析纯化蛋黄多不饱和脂肪酸卵磷脂及其脂肪酸组成分析[J]. 中国食品学报, 2019, 19(6): 267-273. DOI:10.16429/j.1009-7848.2019.06.032.
[38] 周丽媛. 蛋黄卵磷脂的制备及其对代谢综合征小鼠血脂代谢调控机制的研究[D]. 太谷: 山西农业大学, 2022: 15-24. DOI:10.27285/d.cnki.gsxnu.2022.000335.
[39] 严喜鸾, 朱世斌, 李奕潼, 等. 蛋白酶辅助提取卵磷脂工艺优化[J].南昌大学学报(理科版), 2019, 43(3): 251-256. DOI:10.13764/j.cnki.ncdl.2019.03.009.
[40] WANG H, YAO L X, LEE S L, et al. Simultaneous texturization and extraction of phospholipids from liquid egg yolk using renewable solvents[J]. European Journal of Lipid Science and Technology, 2017,119(2): 1500523. DOI:10.1002/ejlt.201500523.
[41] WANG H, YAO L X, LEE S L, et al. Extraction of phospholipids from egg yolk flakes using aqueous alcohols[J]. Journal of the American Oil Chemists’ Society, 2017, 94(2): 309-314. DOI:10.1007/s11746-016-2939-x.
[42] GUO A H, LI S, YANG Y, et al. Lecithin extraction optimisation and synthesis in Hemerocallis citrina Baroni[J]. Scientia Horticulturae,2022, 293: 110682. DOI:10.1016/j.scienta.2021.110682.
[43] PATIL V V, GALGE R V, THORAT B N. Extraction and purification of phosphatidylcholine from soyabean lecithin[J]. Separation and Purification Technology, 2010, 75(2): 138-144. DOI:10.1016/j.seppur.2010.08.006.
[44] SAVOIRE R, SUBRA-PATERNAULT P, BARDEAU T, et al.Selective extraction of phospholipids from food by-products by supercritical carbon dioxide and ethanol and formulating ability of extracts[J]. Separation and Purification Technology, 2020, 238:116394. DOI:10.1016/j.seppur.2019.116394.
[45] GIGI A A, PRAVEENA U, PILLAI P S, et al. Advances and challenges in the fractionation of edible oils and fats through supercritical fluid processing[J]. Comprehensive Reviews in Food Science and Food Safety, 2024, 23(5): e70017. DOI:10.1111/1541-4337.70017.
[46] 高洁, 祁立波, 林松毅, 等. 乙醇对蛋黄磷脂提取纯度及蛋黄粉溶解度的影响[J]. 食品与发酵工业, 2023, 49(7): 212-217. DOI:10.13995/j.cnki.11-1802/ts.031386.
[47] PARK J S, ROY V C, HAN J M, et al. Green extraction of marine phospholipids from Conger eel by-product using SC-CO2: lipidomic profiles and biological activities[J]. Sustainable Chemistry and Pharmacy, 2024, 42: 101742. DOI:10.1016/j.scp.2024.101742.
[48] HAQ M, CHUN B S. Characterization of phospholipids extracted from Atlantic salmon by-product using supercritical CO2 with ethanol as co-solvent[J]. Journal of Cleaner Production, 2018, 178: 186-195.DOI:10.1016/j.jclepro.2018.01.024.
[49] SUBRA-PATERNAULT P, THONGDENG H, GRÉLARD A, et al.Extraction of phospholipids from scallop by-product using supercritical CO2/alcohol mixtures[J]. LWT-Food Science and Technology, 2015,60(2): 990-998. DOI:10.1016/j.lwt.2014.09.057.
[50] KRISHNEGOWDA R, RAVINDRA M R, SHARMA M. Application of supercritical fluid extraction for extraction or enrichment of phospholipids in egg and dairy products: a review[J]. Journal of Food Process Engineering, 2021, 44(6): e13692. DOI:10.1111/jfpe.13692.
[51] ARO H, JÄRVENPÄÄ E P, KÖNKÖ K, et al. Isolation and purification of egg yolk phospholipids using liquid extraction and pilot-scale supercritical fluid techniques[J]. European Food Research and Technology, 2009, 228(6): 857-863. DOI:10.1007/s00217-008-0998-4.
[52] BOT F, COSSUTA D, O’MAHONY J A. Inter-relationships between composition, physicochemical properties and functionality of lecithin ingredients[J]. Trends in Food Science & Technology, 2021, 111: 261-270. DOI:10.1016/j.tifs.2021.02.028.
[53] MANJULA S, KOBAYASHI I, SUBRAMANIAN R. Characterization of phospholipid reverse micelles in nonaqueous systems in relation to their rejection during membrane processing[J]. Food Research International, 2011, 44(4): 925-930. DOI:10.1016/j.foodres.2011.01.059.
[54] LIU F B, LIU Y F, LIU X J, et al. Preparation of deoiled soy lecithin by ultrafiltration[J]. Journal of the American Oil Chemists’ Society,2011, 88(11): 1807-1812. DOI:10.1007/s11746-011-1835-7.
[55] LI B F, CUI Y, JAPIP S, et al. Graphene oxide (GO) laminar membranes for concentrating pharmaceuticals and food additives in organic solvents[J]. Carbon, 2018, 130: 503-514. DOI:10.1016/j.carbon.2018.01.040.
[56] YAN Z Y, ZHENG M Q, YE D H, et al. Reversed micellar-enhanced ultrafiltration removal of oil from concentrated phospholipids[J].Chemical Engineering Science, 2023, 277: 118863. DOI:10.1016/j.ces.2023.118863.
[57] BASSO R C, GONÇALVES L A G, GRIMALDI R, et al. Degumming and production of soy lecithin, and the cleaning of a ceramic membrane used in the ultrafiltration and diafiltration of crude soybean oil[J]. Journal of Membrane Science, 2009, 330(1/2): 127-134.DOI:10.1016/j.memsci.2008.12.052.
[58] CHAVAN P, SHARMA P, SHARMA S R, et al. Application of highintensity ultrasound to improve food processing efficiency: a review[J].Foods, 2022, 11(1): 122. DOI:10.3390/foods11010122.
[59] DENG Y, WANG W J, ZHAO S N, et al. Ultrasound-assisted extraction of lipids as food components: mechanism, solvent, feedstock, quality evaluation and coupled technologies: a review[J]. Trends in Food Science &Technology, 2022, 122: 83-96. DOI:10.1016/j.tifs.2022.01.034.
[60] OPRESCU E E, ENASCUTA C E, RADU E, et al. Does the ultrasonic field improve the extraction productivity compared to classical methods: maceration and reflux distillation?[J]. Chemical Engineering and Processing-Process Intensification, 2022, 179: 109082.DOI:10.1016/j.cep.2022.109082.
[61] HUANG J, LU F Y, WU Y J, et al. Enzymatic extraction and functional properties of phosphatidylcholine from chicken liver[J]. Poultry Science, 2022, 101(6): 101689. DOI:10.1016/j.psj.2021.101689.
[62] DAS S, NADAR S S, RATHOD V K. Integrated strategies for enzyme assisted extraction of bioactive molecules: a review[J]. International Journal of Biological Macromolecules, 2021, 191: 899-917.DOI:10.1016/j.ijbiomac.2021.09.060.
[63] NADAR S S, RAO P, RATHOD V K. Enzyme assisted extraction of biomolecules as an approach to novel extraction technology:a review[J]. Food Research International, 2018, 108: 309-330.DOI:10.1016/j.foodres.2018.03.006.
[64] BARRY K M, DINAN T G, KELLY P M. Pilot scale production of a phospholipid-enriched dairy ingredient by means of an optimised integrated process employing enzymatic hydrolysis, ultrafiltration and super-critical fluid extraction[J]. Innovative Food Science & Emerging Technologies, 2017, 41: 301-306. DOI:10.1016/j.ifset.2017.04.004.
[65] SHINN S, LIYANAGE R, LAY J, et al. Improved fatty acid analysis of conjugated linoleic acid rich egg yolk triacylglycerols and phospholipid species[J]. Journal of Agricultural and Food Chemistry,2014, 62(28): 6608-6615. DOI:10.1021/jf501100y.
[66] STAREK M, HOMA K, STĘPIŃSKA J, et al. Development of thinlayer chromatography densitometry for the quantification of lecithin in dietary supplements[J]. Journal of Planar Chromatography, 2023,36(2): 99-110. DOI:10.1007/s00764-023-00234-3.
[67] 肖虹, 周裔彬, 尚岩岩, 等. 超声辅助提取鸡蛋卵磷脂的工艺研究[J]. 食品工业科技, 2010, 31(8): 255-257. DOI:10.13386/j.issn1002-0306.2010.08.010.
[68] 杨亦文, 李艳, 吴彩娟, 等. HPLC-RI法快速准确测定大豆磷脂酰胆碱含量[J]. 分析测试学报, 2004, 23(5): 118-121.
[69] 孔凡华, 王轩, 于连洋, 等. 高效液相色谱-蒸发光检测器法测定鸡蛋中卵磷脂的含量[J]. 食品工业科技, 2021, 42(13): 299-303.DOI:10.13386/j.issn1002-0306.2020080041.
[70] 王轩, 李全霞, 刘伟, 等. 食品中卵磷脂的检测技术研究进展[J]. 食品与发酵工业, 2018, 44(8): 322-327. DOI:10.13995/j.cnki.11-1802/ts.015186.
[71] TSUGAWA H, IKEDA K, TAKAHASHI M, et al. A lipidome atlas in MS-DIAL 4[J]. Nature Biotechnology, 2020, 38(10): 1159-1163.DOI:10.1038/s41587-020-0531-2.
[72] YEO J, KANG J, KIM H, et al. A critical overview of HPLC-MS-based lipidomics in determining triacylglycerol and phospholipid in foods[J]. Foods, 2023, 12(17): 3177. DOI:10.3390/foods12173177.
[73] HOLČAPEK M, EKROOS K. Mass spectrometry for lipidomics:methods and applications[M]. Hoboken: Wiley, 2023.DOI:10.1002/9783527836512.
[74] ZHANG W P, JIAN R J, ZHAO J, et al. Deep-lipidotyping by mass spectrometry: recent technical advances and applications[J]. Journal of Lipid Research, 2022, 63(7): 100219. DOI:10.1016/j.jlr.2022.100219.
[75] TAKAHASHI R, NAKAYA M, KOTANIGUCHI M, et al.Analysis of phosphatidylethanolamine, phosphatidylcholine, and plasmalogen molecular species in food lipids using an improved 2D high-performance liquid chromatography system[J]. Journal of Chromatography B, Analytical Technologies in the Biomedical and Life Sciences, 2018, 1077/1078: 35-43. DOI:10.1016/j.jchromb.2018.01.014.
[76] SUN Y H, WU Y, FANG B, et al. Comparative analysis of egg yolk phospholipid unsaturation and its impact on neural health in Alzheimer disease mice[J]. Foods, 2025, 14(5): 792. DOI:10.3390/foods14050792.
[77] BAO Z J, ZHANG P L, CHEN J, et al. Egg yolk phospholipids reverse scopolamine-induced spatial memory deficits in mice by attenuating cholinergic damage[J]. Journal of Functional Foods, 2020, 69: 103948.DOI:10.1016/j.jff.2020.103948.
[78] NORRIS G H, PORTER C M, JIANG C, et al. Dietary sphingomyelin attenuates hepatic steatosis and adipose tissue inflammation in high-fatdiet-induced obese mice[J]. The Journal of Nutritional Biochemistry,2017, 40: 36-43. DOI:10.1016/j.jnutbio.2016.09.017.
[79] 尹明雨, 张彩霞, 松冈亮辅, 等. 卵磷脂的生物活性及其应用研究进展[J]. 包装工程, 2020, 41(13): 31-39. DOI:10.19554/j.cnki.1001-3563.2020.13.005.
[80] XIONG W, FANG B, WANG X Y, et al. Dietary lipid intervention in the prevention of brain aging[J]. Engineering, 2024, 37: 128-137.DOI:10.1016/j.eng.2023.04.012.
[81] CONWAY T, SEIDLER K, BARROW M. Unlocking choline’s potential in Alzheimer’s disease: a narrative review exploring the neuroprotective and neurotrophic role of phosphatidylcholine and assessing its impact on memory and learning[J]. Clinical Nutrition ESPEN, 2024, 64: 177-195. DOI:10.1016/j.clnesp.2024.09.024.
[82] MURIEL P, LÓPEZ-SÁNCHEZ P, RAMOS-TOVAR E. Fructose and the liver[J]. International Journal of Molecular Sciences, 2021, 22(13):6969. DOI:10.3390/ijms22136969.
[83] SONG C, LONG X, HE J B, et al. Recent evaluation about inflammatory mechanisms in nonalcoholic fatty liver disease[J].Frontiers in Pharmacology, 2023, 14: 1081334. DOI:10.3389/fphar.2023.1081334.
[84] NORRIS G H, BLESSO C N. Dietary sphingolipids: potential for management of dyslipidemia and nonalcoholic fatty liver disease[J].Nutrition Reviews, 2017, 75(4): 274-285. DOI:10.1093/nutrit/nux004.
[85] SÁNCHEZ V, BAUMANN A, BRANDT A, et al. Oral supplementation of phosphatidylcholine attenuates the onset of a dietinduced metabolic dysfunction-associated steatohepatitis in female C57BL/6J mice[J]. Cellular and Molecular Gastroenterology and Hepatology, 2024, 17(5): 785-800. DOI:10.1016/j.jcmgh.2024.01.009.
[86] CHOI J, SONG I, LEE S M, et al. Protective effects of phosphatidylcholine against hepatic and renal cell injury from advanced glycation end products[J]. Medicina, 2022, 58(11): 1519.DOI:10.3390/medicina58111519.
[87] LI Y M, TIAN X T, LUO J Y, et al. Molecular mechanisms of aging and anti-aging strategies[J]. Cell Communication and Signaling, 2024,22(1): 285. DOI:10.1186/s12964-024-01663-1.
[88] CASARES D, ESCRIBÁ P V, ROSSELLÓ C A. Membrane lipid composition: effect on membrane and organelle structure, function and compartmentalization and therapeutic avenues[J]. International Journal of Molecular Sciences, 2019, 20(9): 2167. DOI:10.3390/ijms20092167.
[89] SUN N, CHEN J, BAO Z J, et al. Egg yolk phosphatidylethanolamine:extraction optimization, antioxidative activity, and molecular structure profiling[J]. Journal of Food Science, 2019, 84(5): 1002-1011.DOI:10.1111/1750-3841.14512.
[90] CHEN Y L, CHEN K, ZHU H, et al. Methyltransferase Setd2 prevents T cell-mediated autoimmune diseases via phospholipid remodeling[J].PNAS2024, 121(8): e2314561121. DOI:10.1073/pnas.2314561121.
[91] AZARCOYA-BARRERA J, WOLLIN B, VEIDA-SILVA H, et al.Egg-phosphatidylcholine attenuates T-cell dysfunction in high-fat diet fed male Wistar rats[J]. Frontiers in Nutrition, 2022, 9: 811469.DOI:10.3389/fnut.2022.811469.
[92] RUSNAK T, AZARCOYA-BARRERA J, WOLLIN B, et al. A physiologically relevant dose of 50% egg-phosphatidylcholine is sufficient in improving gut permeability while attenuating immune cell dysfunction induced by a high-fat diet in male Wistar rats[J].The Journal of Nutrition, 2023, 153(10): 3131-3143. DOI:10.1016/j.tjnut.2023.08.010.
[93] ATIK D S, BÖLÜK E, TOKER O S, et al. Investigating the effects of Lecithin-PGPR mixture on physical properties of milk chocolate[J].LWT-Food Science and Technology, 2020, 129: 109548. DOI:10.1016/j.lwt.2020.109548.
[94] PANPIPAT W, CHUMIN T, THONGKAM P, et al. Relatively low lecithin inclusion improved gelling characteristics and oxidative stability of single-washed mackerel (Auxis thazard) surimi[J]. Foods,2024, 13(4): 546. DOI:10.3390/foods13040546.
[95] ZHOU X X, LIN H H, ZHU S C, et al. Textural, rheological and chemical properties of surimi nutritionally-enhanced with lecithin[J].LWT-Food Science and Technology, 2020, 122: 108984. DOI:10.1016/j.lwt.2019.108984.
[96] SHI T, WIJAYA G Y A, YUAN L, et al. Gel properties of Amur sturgeon (Acipenser schrenckii) surimi improved by lecithin at reduced and regular-salt concentrations[J]. RSC Advances, 2020, 10(51):30896-30906. DOI:10.1039/D0RA04487C.
[97] AHMED T B, EGGESBØ M, CRISWELL R, et al. Total fatty acid and polar lipid species composition of human milk[J]. Nutrients, 2022,14(1): 158. DOI:10.3390/nu14010158.
[98] YU X X, ZHAO Y J, SUN M, et al. Effects of egg yolk lecithin/milk fat globule membrane material ratio on the structure and stability of oil-in-water emulsions[J]. LWT-Food Science and Technology, 2022,168: 113891. DOI:10.1016/j.lwt.2022.113891.
[99] PAN Y, ZHANG L, FU X F, et al. Addition of phospholipids improved the physical stability and fat globule structure of processed milk[J].Foods, 2025, 14(3): 375. DOI:10.3390/foods14030375.
[100] MA Q, SUN M, ZHAO Y J, et al. Improving lipid digestion by modulating interfacial structure of fat globule based on milk fat globule membrane and different phospholipids[J]. Food Hydrocolloids,2024, 150: 109736. DOI:10.1016/j.foodhyd.2024.109736.
[101] YU X X, ZHOU W L, JIA Z B, et al. Interfacial composition in infant formulas powder modulate lipid digestion in simulated in-vitro infant gastrointestinal digestion[J]. Food Research International, 2023, 165:112553. DOI:10.1016/j.foodres.2023.112553.
[102] CHEN D W, BALAGIANNIS D P, PARKER J K. Egg yolk phospholipids: a functional food material to generate deep-fat frying odorants[J]. Journal of the Science of Food and Agriculture, 2019,99(14): 6638-6643. DOI:10.1002/jsfa.9939.
[103] CHEN D W, BALAGIANNIS D P, PARKER J K. Use of egg yolk phospholipids to generate chicken meat odorants[J]. Food Chemistry,2019, 286: 71-77. DOI:10.1016/j.foodchem.2019.01.184.
[104] CHEN D W, WAN P, YAO J Y, et al. Egg yolk phospholipids as an ideal precursor of fatty note odorants for chicken meat and fried foods: a review[J]. Food Chemistry, 2023, 407: 135177. DOI:10.1016/j.foodchem.2022.135177.
[105] CASTRO-GÓMEZ P, GARCIA-SERRANO A, VISIOLI F, et al.Relevance of dietary glycerophospholipids and sphingolipids to human health[J]. Prostaglandins, Leukotrienes, and Essential Fatty Acids,2015, 101: 41-51. DOI:10.1016/j.plefa.2015.07.004.
[106] AJEESHKUMAR K K, ANEESH P A, RAJU N, et al. Advancements in liposome technology: preparation techniques and applications in food, functional foods, and bioactive delivery: a review[J].Comprehensive Reviews in Food Science and Food Safety, 2021,20(2): 1280-1306. DOI:10.1111/1541-4337.12725.
[107] EMAMI S, AZADMARD-DAMIRCHI S, PEIGHAMBARDOUST S H, et al. Liposomes as carrier vehicles for functional compounds in food sector[J]. Journal of Experimental Nanoscience, 2016, 11(9):737-759. DOI:10.1080/17458080.2016.1148273.
[108] AKRAM N, AFZAAL M, SAEED F, et al. Liposomes: a promising delivery system for active ingredients in food and nutrition[J].International Journal of Food Properties, 2023, 26(1): 2476-2492.DOI:10.1080/10942912.2023.2247578.
[109] BONDU C, YEN F T. Nanoliposomes, from food industry to nutraceuticals: interests and uses[J]. Innovative Food Science &Emerging Technologies, 2022, 81: 103140. DOI:10.1016/j.ifset.2022.103140.
[110] LEE M K. Liposomes for enhanced bioavailability of water-insoluble drugs: in vivo evidence and recent approaches[J]. Pharmaceutics, 2020,12(3): 264. DOI:10.3390/pharmaceutics12030264.
[111] PATIL J, PAWDE D M, BHATTACHARYA S, et al. Phospholipid complex formulation technology for improved drug delivery in oncological settings: a comprehensive review[J]. AAPS PharmSciTech,2024, 25(5): 91. DOI:10.1208/s12249-024-02813-x.
[112] DYMEK M, SIKORA E. Liposomes as biocompatible and smart delivery systems: the current state[J]. Advances in Colloid and Interface Science, 2022, 309: 102757. DOI:10.1016/j.cis.2022.102757.
[113] ZHANG N, SONG J, HAN Y C. Research progress of phospholipid vesicles in biological field[J]. Biomolecules, 2024, 14(12): 1628.DOI:10.3390/biom14121628.
[114] BRÜGGEMANN R J, JENSEN G M, LASS-FLÖRL C. Liposomal amphotericin B: the past[J]. Journal of Antimicrobial Chemotherapy,2022, 77(Suppl 2): ii3-ii10. DOI:10.1093/jac/dkac351.
[115] GADSDEN J, HAMILTON M, SCHWARTZ G, et al. A phase 3 active-controlled trial of liposomal bupivacaine via adductor canal block for total knee arthroplasty[J]. The Journal of Arthroplasty, 2025,40(10): 2605-2614.e5. DOI:10.1016/j.arth.2025.07.030.
[116] VAN HOOGEVEST P, FAHR A. Phospholipids in cosmetic carriers[M]// CORNIER J, KECK C M, VAN DE VOORDE.Nanocosmetics, Cham: Springer, 2019: 95-140. DOI:10.1007/978-3-030-16573-4_6.
[117] DYMEK M, OLECHOWSKA K, HĄC-WYDRO K, et al.Liposomes as carriers of GHK-Cu tripeptide for cosmetic application[J]. Pharmaceutics, 2023, 15(10): 2485. DOI:10.3390/pharmaceutics15102485.
[118] KARKAD A A, PIRKOVIĆ A, MILOŠEVIĆ M, et al. Silibininloaded liposomes: the influence of modifications on physicochemical characteristics, stability, and bioactivity associated with dermal application[J]. Pharmaceutics, 2024, 16(11): 1476. DOI:10.3390/pharmaceutics16111476.
[119] JOVANOVIĆ A A, BALANČ B, VOLIĆ M, et al. Rosehip extractloaded liposomes for potential skin application: physicochemical properties of non- and UV-irradiated liposomes[J]. Plants, 2023,12(17): 3063. DOI:10.3390/plants12173063.
[120] CHAVES M A, FERREIRA L S, BALDINO L, et al. Current applications of liposomes for the delivery of vitamins: a systematic review[J]. Nanomaterials, 2023, 13(9): 1557. DOI:10.3390/nano13091557.
[121] WANG M Z, YAN W Q, ZHOU Y L, et al. Progress in the application of lecithins in water-in-oil emulsions[J]. Trends in Food Science &Technology, 2021, 118: 388-398. DOI:10.1016/j.tifs.2021.10.019.
[122] KIM E J, KONG B J, KWON S S, et al. Preparation and characterization of W/O microemulsion for removal of oily make-up cosmetics[J]. International Journal of Cosmetic Science, 2014, 36(6):606-612. DOI:10.1111/ics.12163.